MedPacto, an innovative new drug development company, announced on November 10 that it has achieved a complete response (CR) in its clinical trial of "Vactosertib" for osteosarcoma.
MedPacto presented interim results from its global Phase 1 clinical trial of the immuno-oncology candidate "Vactosertib" for osteosarcoma patients at the annual meeting of the Society for Immunotherapy of Cancer (SITC 2025), which was held in Maryland, United States, from November 7 to 9.
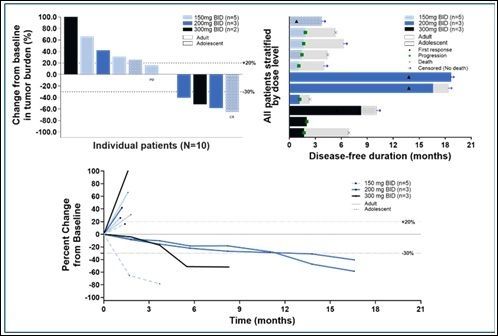
In a study involving 11 patients with relapsed or refractory (R/R) osteosarcoma who received "Vactosertib" as a monotherapy, one patient who had experienced multiple recurrences despite various prior treatments achieved a complete response (CR). Three patients showed a partial response (PR). The objective response rate (ORR) was 36.4%, more than three times higher than existing therapies. The safety and tolerability profiles were also excellent, and no dose-limiting toxicity (DLT) was observed.
MedPacto explained that achieving a complete response with a high trend of efficacy in the relapsed and refractory osteosarcoma patient group is an extremely rare case worldwide.
MedPacto attracted significant attention even before the opening of SITC by being selected for the "Late Breaking Abstract (LBA)" track. The LBA is known to be reserved for innovative achievements based on the latest clinical data.
The clinical trial of Vactosertib for osteosarcoma is currently in Phase 2 for adult patients, and the second cohort of Phase 1 for adolescent patients is also underway.
A MedPacto representative stated, "Through this presentation, we have proven that Vactosertib demonstrates high efficacy in osteosarcoma even as a monotherapy," adding, "As the enrollment for the Phase 2 trial in adults is progressing smoothly, we will do our best to quickly secure Phase 2 data and achieve commercial success."
Osteosarcoma is a rare malignant tumor that occurs in bone and cartilage. Combination chemotherapy is the standard treatment. However, due to limitations in improving treatment efficacy and survival rates, the global market is expected to expand significantly if a new therapy is developed.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.