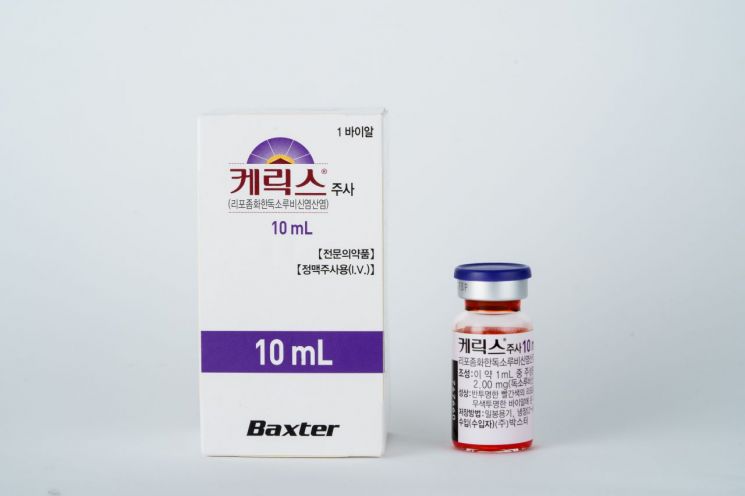
[Asia Economy Reporter Lee Gwan-joo] Chong Kun Dang announced on the 23rd that it has signed an exclusive domestic sales contract for Baxter Korea's recurrent ovarian cancer treatment drug, Kerix.
Under this contract, Chong Kun Dang will be responsible for the distribution, sales, and marketing of Kerix at domestic hospitals and clinics starting in July.
Kerix's main ingredient, doxorubicin, is encapsulated in a pegylated liposome using pegylation platform technology, which targets delivery specifically to cancer cells. Compared to conventional doxorubicin drugs, it features reduced side effects such as cardiotoxicity and hair loss, and has a longer-lasting effect.
Approved by the U.S. Food and Drug Administration (FDA) in 1995, it has been used for ovarian cancer patients in global markets including the U.S. and Europe. Last year, it was recommended as a first-line therapy for platinum-sensitive and resistant recurrent ovarian cancer patients in the National Comprehensive Cancer Network (NCCN) guidelines.
A Chong Kun Dang official stated, "We will strategically carry out the distribution, sales, and marketing of Kerix by actively utilizing the sales capabilities and know-how accumulated through our self-developed ovarian cancer drug, Camtobel. We plan to further strengthen our position in the recurrent ovarian cancer treatment market through continuous cooperation with Baxter Korea."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.