FDA Expands Vaccination Eligibility
Goal to Normalize Fall Semester Classes
MFDS "Proceeding with Approval Changes Promptly"
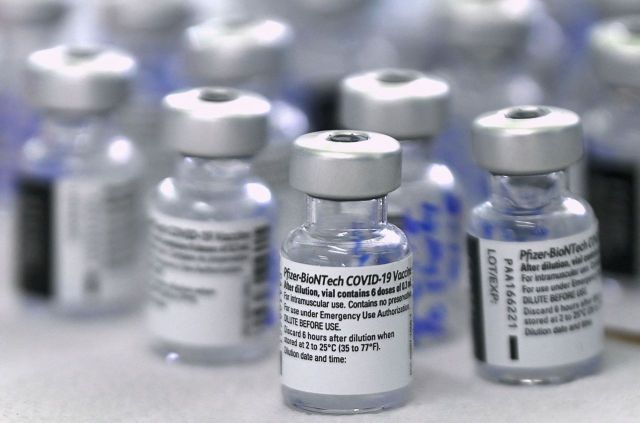
[Asia Economy reporters Seo So-jeong and Cho Hyun-ui] The United States has expanded the authorized use of Pfizer's COVID-19 vaccine to individuals aged 12 and older.
According to the U.S. economic media CNBC on the 10th (local time), the U.S. Food and Drug Administration (FDA) has decided to allow the Pfizer COVID-19 vaccine to be administered to minors aged 12 to 15. Accordingly, the vaccination age has been lowered from 16 and older to 12 and older. Janet Woodcock, Acting FDA Commissioner, stated, "This approval allows a younger age group to be protected from COVID-19," adding, "The return to normal life and the end of the pandemic are now closer."
The vaccination is expected to begin after the Advisory Committee on Immunization Practices (ACIP), a CDC advisory body, issues its recommendation on this measure on the 12th. The Biden administration aims to fully normalize classes starting in the fall semester. CNBC reported, "State governments will begin vaccinating students before the fall semester," and added, "This will be a positive signal not only for the normalization of classes but also for the resumption of after-school activities."
Canada expanded the authorized use of Pfizer's vaccine to those aged 12 and older on the 5th. The European Medicines Agency (EMA) is expected to announce its decision on allowing use for those aged 12 and older next month. Pfizer is also testing vaccines for children aged 6 months to 11 years. The Hill, a U.S. congressional publication, reported, "Pfizer is expected to request approval for COVID-19 vaccine use for children aged 2 to 11 around September."
South Korea is also preparing to expand the vaccination age group. A Pfizer Korea official stated, "As the vaccine approval target has expanded in the U.S., we are currently preparing data to request a change in approval from the domestic Ministry of Food and Drug Safety (MFDS)," adding, "We will apply soon." The MFDS plans to proceed with follow-up procedures once the approval change application is submitted by the company.
When the MFDS approved Pfizer's vaccine 'Comirnaty' in March, it authorized use for those aged 16 and older. Park In-sook, head of the Biologics Evaluation Division at the MFDS, said, "Since emergency use was approved in the U.S. and Canada has already lowered the vaccination age, approval is expected to be possible soon."
President Moon Jae-in emphasized the day before, "We will continue efforts to secure additional supplies considering the possibility of a third dose, preparation against variant viruses, expansion of vaccination targets to minors and children, and the quantity needed next year." According to the Korea Disease Control and Prevention Agency, high school seniors and their teachers will receive COVID-19 vaccinations during this summer vacation. The number of high school seniors to be vaccinated is estimated to be between 450,000 and 490,000.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.