The World's Only Subcutaneous Infliximab Injection
Listed Through Contract with Top 3 Prescription Drug Reimbursement Companies
Seo Jung-jin: "Annual Sales Potential at Least 5 Trillion Won"
Celltrion's new autoimmune disease treatment drug, Jimpendra (the U.S. launch name for Remsima SC), is accelerating its entry into the U.S. market by successfully targeting major U.S. pharmacy benefit managers (PBMs).
On the 5th, Celltrion announced on its website that it had signed formulary listing contracts with all three major U.S. PBMs: CVS Caremark, Express Scripts, and OptumRx. Following the first contract with Express Scripts in April, the company announced on the 1st that it had signed a contract with the second of the three major PBMs, and the very next day, "on the 2nd (local time), the contract for Jimpendra listing was completed with the remaining one of the three major U.S. PBMs," the company explained.
Earlier, Kim Hyung-ki, Vice Chairman of Celltrion, had promised, "We will be able to list on all three major PBMs within this year." This achievement reflects the company's determination not to repeat the difficulties faced by other biosimilars in securing PBM contracts during their initial U.S. market entry, which had hindered early market share growth.
However, Celltrion did not disclose the specific PBM involved in the last contract and explained that, as with the previous contract, "due to contractual terms, it is difficult to specify the PBM at this time." Nonetheless, since contracts have been secured with all three major PBMs, there is expected to be no particular doubt in the market.
PBMs, the 'First Gateway' to Market Penetration... Enabling Access to Most U.S. Patients
Jimpendra is intensifying its U.S. market penetration by leveraging its convenience as the "world's only subcutaneous infliximab injection." The active ingredient in Jimpendra, infliximab, has proven efficacy and safety for inflammatory bowel diseases such as ulcerative colitis and Crohn's disease. Existing drugs with this ingredient have only been available as intravenous injections administered at hospitals or clinics. Given the difficulty of visiting medical institutions in the U.S., there has been strong market demand for a subcutaneous injection formulation that patients can self-administer.
For practical market penetration, listing on PBMs is essential. In the U.S. pharmaceutical market, drugs must be included in the "formulary," a list of drugs managed by PBMs on behalf of public and private insurers, to be realistically sold. If Jimpendra is not on the formulary of the patient's insurance provider, the patient would have to bear the full cost of the drug themselves.
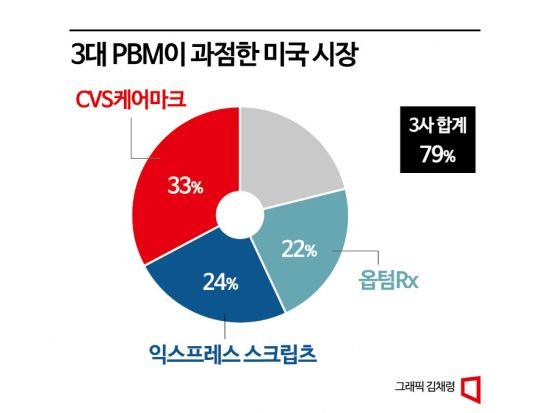
Currently, the U.S. PBM market is dominated by these three major PBMs, which collectively hold about 80% market share. However, the U.S. insurance market is divided between private insurance and public insurance such as Medicare and Medicaid. While the second contract with a large PBM covered both public and private insurance, the third contract with another large PBM covers only the public insurance market, so it does not yet cover the full 80%. Nevertheless, when combined with contracts with other small and medium-sized PBMs, Jimpendra can access about 75% of the U.S. insurance market just five months after its launch. The company also stated that "only private insurance contracts remain to be negotiated with the third large PBM."
Since June, Jimpendra has been receiving insurance reimbursements through contracts with PBMs. Even before reimbursement, the company supplied the product free of charge through a start program to patients enrolled in PBMs where formulary listing had not yet been completed, or doctors applied for prior authorization from insurers based on patient condition and provided support without reimbursement. This has laid the foundation for actual sales expansion.
According to IQVIA, a pharmaceutical market research firm, the inflammatory bowel disease market in the U.S., where Jimpendra is currently focused, is estimated to be worth $10.3 billion (approximately 14 trillion KRW). Celltrion plans to secure more than 10% market share by next year. Beyond that, if it succeeds in accelerating sales by increasing the conversion rate to the subcutaneous injection formulation and the number of registered patients, annual sales exceeding 2 trillion KRW are expected. Seo Jung-jin, Chairman of Celltrion Group, who is directly overseeing Jimpendra's U.S. market entry while residing in the U.S., has expressed confidence in Jimpendra, stating, "It can generate annual sales of up to 3 trillion KRW within three years," and "Even conservatively, it could reach up to 5 trillion KRW."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














