Blockbuster Drug Patents Expire One After Another
Development Accelerates for Injectables Like 'Ailia'
The scope of biosimilar entry in the domestic pharmaceutical industry is expanding to ophthalmic diseases. As blockbuster drugs for macular degeneration are consecutively losing their patents or are about to expire, biosimilar developers are stepping up to the challenge. Biosimilars, which were mainly focused on autoimmune diseases and anticancer drugs, are now opening their eyes to a new frontier in ophthalmic diseases.
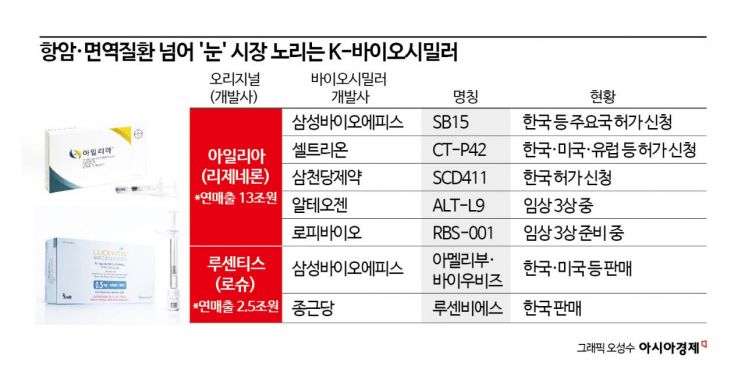
On the 1st, Samsung Bioepis signed a domestic sales contract with Samil Pharmaceutical for ‘SB15,’ a biosimilar developed for the wet macular degeneration treatment drug ‘Eylea.’ Samsung Bioepis also entrusts the sales of ‘Amelibu,’ a biosimilar of another macular degeneration drug ‘Lucentis,’ to Samil Pharmaceutical. Samsung’s financially backed macular degeneration biosimilars are being sold through the sales power of Samil Pharmaceutical, a leading domestic ophthalmology-specialized pharmaceutical company.
Macular degeneration is a representative cause of blindness. Blood and other fluids leaking from abnormally grown capillaries in the macula, the central part of the retina, adhere to the retina and obscure vision. Drugs like Eylea and Lucentis, which inhibit the formation of new blood vessels, must be injected regularly to suppress progression. Aging is a major cause, and with the aging population, the incidence is rapidly increasing worldwide. Accordingly, the global macular degeneration treatment market is expected to grow at an average annual rate of 8%, reaching $16.3 billion (approximately 22 trillion KRW) by 2027. Eylea and Lucentis are already global blockbuster drugs with sales of $9.65 billion (about 13 trillion KRW) and $1.87 billion (about 2.5 trillion KRW) respectively in 2022.
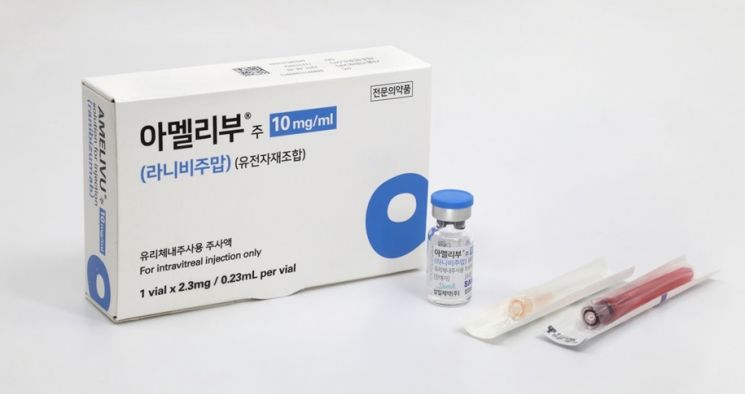 Samsung Bioepis's biosimilar for macular degeneration treatment, 'Amelibu'
Samsung Bioepis's biosimilar for macular degeneration treatment, 'Amelibu' [Photo provided by Samil Pharmaceutical]
The biosimilar competition for these two drugs has intensified recently because their patents have expired. Lucentis’ patent has already expired, and Eylea’s domestic patent expired on the 9th. In the U.S., the patent will expire in May, and in Europe, in November next year. Since Eylea is a protein-based biologic and Lucentis is an antibody-based biologic, they must be produced as biosimilars rather than generics, which are copies of synthetic drugs. Unlike synthetic drugs, which can be exactly replicated according to the original blueprint, it is impossible to create an identical biologic drug, making the development process much more difficult.
Domestic biosimilar developers are focusing on Eylea. In addition to Samsung Bioepis, Celltrion, Samchundang Pharmaceutical, Alteogen, and Rophibio have started development. On the other hand, only Samsung Bioepis and Chong Kun Dang have developed biosimilars for Lucentis. This is because Eylea dominates the market due to its high dosing convenience. Both drugs are injected directly into the eye. Lucentis requires monthly injections, but Eylea is administered every two months, making it more preferred by patients.
Domestic Eylea biosimilar developers are accelerating their final development stages. Samsung Bioepis is undergoing approval procedures for SB15 in Korea and other major countries. Celltrion applied for approval of ‘CT-P42’ last year in Korea, the U.S., and Europe. Samchundang Pharmaceutical submitted an approval application for ‘SCD411’ to the Korean Ministry of Food and Drug Safety. Alteogen plans to complete clinical trials for ‘ALT-L9’ within this year. Rophibio also applied for approval of its phase 3 clinical trial in Korea at the end of last year.
Meanwhile, original developers continue to release new drugs to prevent losing market share to biosimilars. Regeneron, the developer of Eylea, received approval from the U.S. Food and Drug Administration (FDA) last year for ‘EyleaHD,’ which increased the dosage from 2 mg to 8 mg. The dosing interval was doubled from 8 weeks to 16 weeks while maintaining efficacy. Roche, the developer of Lucentis, introduced a new drug called ‘Vabysmo,’ which inhibits new blood vessel formation while stabilizing existing retinal vessels. Although injections are required monthly for the first four months of treatment, subsequent injections can be administered every 8 to 16 weeks. Clinical trials confirmed that its vision improvement effect is not inferior to Eylea. Vabysmo received FDA approval in 2022 and has been covered by domestic health insurance since October last year.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














