New Drug for Diffuse Large B-Cell Lymphoma
Roche's rare blood cancer treatment has received import approval in South Korea.
The Ministry of Food and Drug Safety announced on the 7th that it has approved Korean Roche's diffuse large B-cell lymphoma treatment 'Columvi (active ingredient glofitamab)'.
Diffuse large B-cell lymphoma, a rare blood cancer, is one of the most common lymphomas and is characterized by rapid progression.
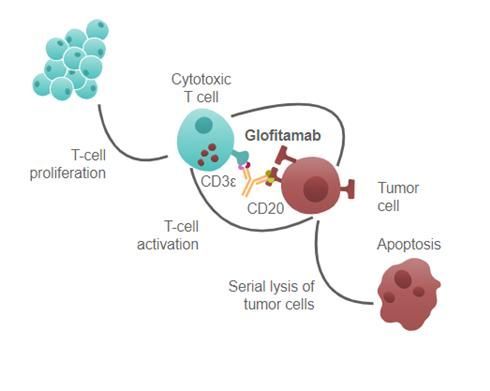
Columvi is a CD20/CD3 bispecific monoclonal antibody that binds to CD3 on the surface of T cells and CD20 on the surface of B cells. When this drug binds to CD3, immune cells called T cells are activated. Then, by also binding to CD20, it positions the B cells next to the activated T cells, inducing the lysis of B cells.
Columvi can be used for patients with relapsed or refractory diffuse large B-cell lymphoma after two or more systemic therapies.
An official from the Ministry of Food and Drug Safety stated, "We will continue to do our best to ensure that treatments with sufficiently confirmed safety and efficacy are supplied promptly, based on regulatory science expertise."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














