Global 42 Trillion Won Gastroesophageal Reflux Disease Market
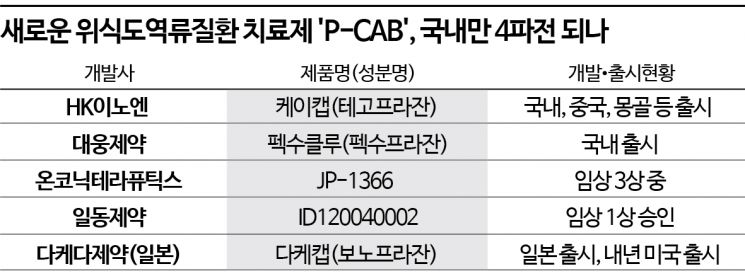
4-Way Competition in Korea as Ildong Pharmaceutical Joins Development
HK Innoen's 'K-CAB' and Daewoong's 'Pexuclu' Rapidly Dominate Market
Jeil's 'JP-1366' Expected to Complete Clinical Trials Within the Year
[Asia Economy Reporter Lee Chun-hee] In the gastroesophageal reflux disease (GERD) treatment market, the share of 'Potassium-Competitive Acid Blockers (P-CAB)' is rapidly growing, prompting various pharmaceutical companies to launch challenges one after another. Since overseas pharmaceutical companies' development is still insufficient, domestic pharmaceutical companies are quickly dominating the overseas market as well.
According to the industry on the 16th, recently Ildong Pharmaceutical received approval from the Ministry of Food and Drug Safety for the Phase 1 clinical trial plan (IND) of 'ID120040002,' a P-CAB mechanism drug. This is a stepwise dose-escalation clinical trial to evaluate the safety of ID120040002 in healthy adult males. The trial will be conducted at Seoul National University Hospital.
P-CAB formulations are gaining attention in the GERD treatment field due to various advantages compared to the existing treatment, 'Proton Pump Inhibitors (PPI).' PPIs must be taken 30 minutes before meals and have inconveniences such as acid secretion occurring at night due to stimulation of acid secretion. Side effects like osteoporosis and stroke have also been controversial. However, P-CAB formulations can be taken regardless of meal times and have improved nighttime heartburn.
The challenge by Ildong Pharmaceutical to develop P-CAB formulations is analyzed as an attempt to replace the existing portfolio as the PPI market gradually shrinks. Since this year, Ildong Pharmaceutical has acquired the domestic distribution rights for AstraZeneca's (AZ) original PPI drug 'Nexium' and started sales activities. Nexium is a global blockbuster drug that has recorded the highest cumulative prescriptions worldwide for about 10 years. Until now, Daewoong Pharmaceutical held the distribution rights and sold it domestically.
However, Nexium's market share has recently been declining. Last year, Nexium's outpatient prescription sales were 48.2 billion KRW, down 13% from the previous year. The top prescription title was taken by Hanmi Pharmaceutical's 'Esmezol,' which grew 22% to 53.8 billion KRW. In addition, after Nexium's patent expiration, various generic products have been sold, leading to competition among various PPI drugs for market share.
On the other hand, P-CAB formulations are rapidly growing, with a total of five substances either launched or entering full development stages domestically. Although Yuhan Corporation's 'Revanex' (active ingredient Levaprazan), developed as the world's first P-CAB drug in 2005, did not achieve significant results, HK Inno.N's 'K-CAB' (Tegoprazan) obtained approval in 2018, followed by Daewoong Pharmaceutical's 'Pexuclu' (Pexuprazan) receiving approval last year, allowing P-CAB formulations to quickly dominate the market.
K-CAB surpassed 100 billion KRW in sales last year, three years after its launch, and is expected to reach 120 billion KRW this year. It has secured various indications including erosive esophagitis, non-erosive reflux disease, gastric ulcers, Helicobacter pylori eradication, and maintenance therapy after erosive GERD treatment. Pexuclu has also settled in the market, recording 4.5 billion KRW in sales in the third quarter since its launch in July.
Additionally, Onconic Therapeutics, a subsidiary of Jeil Pharmaceutical, is conducting Phase 3 clinical trials for 'JP-1366.' The reflux esophagitis indication is expected to complete clinical trials within this year, and the Phase 3 IND for gastric ulcers was approved in June.
 Daewoong Pharmaceutical's gastroesophageal reflux disease treatment 'Pexuclu' (Photo by Daewoong Pharmaceutical)
Daewoong Pharmaceutical's gastroesophageal reflux disease treatment 'Pexuclu' (Photo by Daewoong Pharmaceutical)
As the global GERD treatment market, known to be worth about 42 trillion KRW, undergoes a paradigm shift to P-CAB, there are expectations that domestic companies will dominate the global market. Among multinational pharmaceutical companies, only Japan's Takeda Pharmaceutical has released a P-CAB formulation.
Takeda's 'Takecab' (Vonoprazan) received domestic approval in 2019 but has not been launched since, whereas it is accelerating market entry in the United States. After receiving FDA approval for Helicobacter pylori eradication therapy, it has also applied for FDA approval as a treatment for erosive esophagitis (EE) and received a review deadline of January 11 next year. Additionally, it is conducting Phase 3 clinical trials for non-erosive reflux disease (NERD) and plans to complete the trials next year.
Domestic pharmaceutical companies are also hastening overseas expansion. HK Inno.N started sales of K-CAB in China and Mongolia and last month received FDA approval for a Phase 3 clinical trial IND for GERD in the United States through its US partner Braintree. Daewoong Pharmaceutical also plans to start Phase 3 trials in the US next year with its US partner Neurogastrics. Jeil Pharmaceutical is pursuing Phase 3 trials in Europe.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














