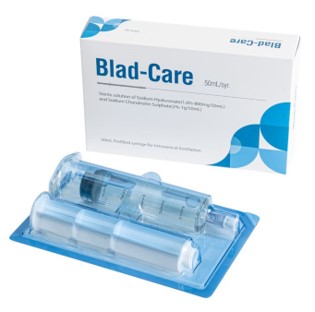
[Asia Economy Reporter Jang Hyowon] BioPlus (CEO Jeong Hyungyu) announced on the 30th that it has signed a supply contract and completed the first shipment of its cystitis treatment 'Blad-Care,' which received product approval from the Ministry of Food and Drug Safety in November, with BioGino Korea.
BioPlus's cystitis treatment ‘Blad-Care’ contains hyaluronic acid (HA) and chondroitin sulfate (CS), biomaterials that are major components of the Glycosaminoglycans (GAGs) layer of bladder epithelial cells. It is a Class 4 medical device that improves symptoms of chronic cystitis and interstitial cystitis by replenishing and protecting the damaged GAGs layer.
BioGino Korea is a healthcare company founded by obstetrics and gynecology specialists and has a network of specialized obstetrics and gynecology hospitals and clinics in Korea. Through this sales contract and first shipment, BioPlus has officially started sales in the domestic urology and obstetrics and gynecology sectors.
A BioPlus official stated, “The global market for cystitis treatments is estimated at about 5 trillion KRW, and the domestic market is about 50 billion KRW. The launch of ‘Blad-Care,’ originally scheduled for the first quarter of next year, was completed within this year to meet market demands. Along with fillers, adhesion prevention agents, and synovial fluid, cystitis treatments are expected to become a new growth item for our company.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














