Vaccine Safety Confirmed... "Monitoring Needed for Individuals with Anaphylaxis History"
[Asia Economy Reporter Seo Sojeong] Pfizer's 'Comirnaty' has received product approval from the Ministry of Food and Drug Safety (MFDS). It is the second COVID-19 vaccine to be approved after AstraZeneca, and the first messenger ribonucleic acid (mRNA) vaccine to be authorized in South Korea.
On the 5th, Kim Gang-rip, Commissioner of the MFDS, stated in a briefing, "On the 5th, a final inspection committee including external experts was held, and the product approval was decided for the COVID-19 vaccine ‘Comirnaty’, imported by Pfizer Korea, which applied for import product approval on January 25, on the condition of submitting the final clinical trial report."
Previously, the MFDS received advisory opinions from the verification advisory group on the 22nd of last month and the Central Pharmaceutical Affairs Deliberation Committee on the 25th of the same month. The final product approval was decided at today's meeting, the last stage of the approval review process. The meeting held this morning was attended by three external experts, including Oh Il-hwan, chairman of the Central Pharmaceutical Affairs Deliberation Committee, and five internal members including the MFDS Commissioner.
Commissioner Kim said, "The final inspection committee judged that it is appropriate to approve the vaccine for those aged 16 and over, consistent with the results of two previous advisory meetings regarding the efficacy and effectiveness for those aged 16 and above."
The final inspection committee explained that it considered ▲the clinical trial was designed to confirm safety and efficacy in subjects aged 16 and older, confirming preventive effects ▲the immune response of adolescents aged 16 and older is expected to be similar to that of adults ▲clinical trial data from adults is available ▲and that many countries including the United States, Europe (EU), the United Kingdom, and Japan have approved it for those aged 16 and above.
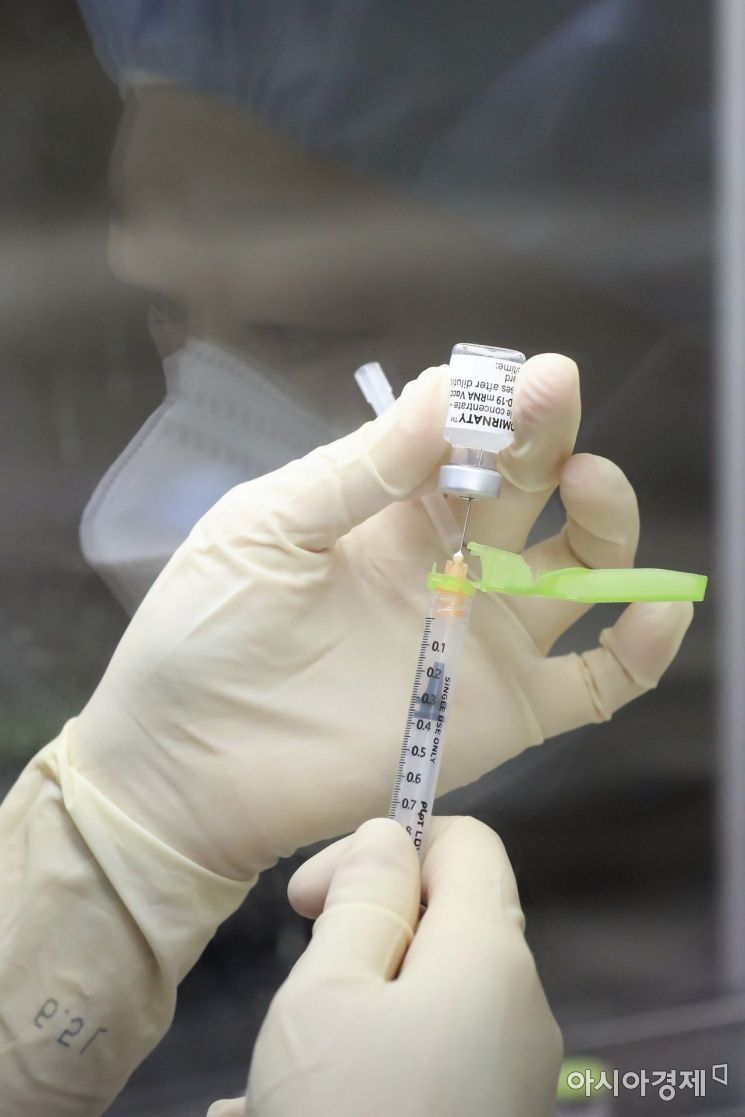 On the morning of the 27th, when Pfizer vaccine administration began for medical staff treating COVID-19 patients, medical personnel were preparing doses of the Pfizer vaccine in syringes at a sterile workstation (clean bench) inside the Central Vaccination Center at the National Medical Center in Jung-gu, Seoul. Photo by Joint Press Corps
On the morning of the 27th, when Pfizer vaccine administration began for medical staff treating COVID-19 patients, medical personnel were preparing doses of the Pfizer vaccine in syringes at a sterile workstation (clean bench) inside the Central Vaccination Center at the National Medical Center in Jung-gu, Seoul. Photo by Joint Press Corps
◆First mRNA vaccine approved domestically= Comirnaty is a messenger ribonucleic acid (mRNA) vaccine jointly developed by Pfizer in the United States and BioNTech in Germany. It injects the COVID-19 virus antigen gene in the form of mRNA, synthesizes antigen proteins in the body, and induces the production of neutralizing antibodies. When the COVID-19 virus invades the human body, these antibodies neutralize and eliminate the virus.
The efficacy and effectiveness of this drug is prevention of COVID-19 in individuals aged 16 and older. After dilution, 0.3 mL is administered once, followed by a booster dose three weeks later. The storage condition is between -60 to -90℃ for six months. Comirnaty is the first mRNA vaccine to be approved domestically and has received conditional approval or emergency use authorization in 59 countries including Europe (EMA), the United States, Japan, and from the World Health Organization (WHO).
The final inspection committee judged that most of the reported adverse events related to safety were expected and generally mild.
Commonly reported adverse events included injection site pain, fever, fatigue, chills, headache, and muscle pain. Symptoms were mostly mild to moderate and resolved within a few days after vaccination. One case of drug-related hypersensitivity (urticaria) occurred after vaccination, and no drug-related anaphylaxis was reported during the clinical trial period.
Among all 43,448 registered clinical trial participants, serious adverse events were reported in 0.6% (126) of the vaccine group and 0.5% (111) of the control group. Of these, four cases of serious adverse drug reactions possibly related to vaccination, including shoulder injuries, were reported. Lymphadenopathy and ventricular arrhythmia have recovered, while shoulder injury, lower back pain, and bilateral lower limb pain are still recovering.
The final inspection committee also judged the vaccine's preventive effect to be sufficient. Clinical trial results showed that 8 people in the vaccine group and 162 in the control group were diagnosed with COVID-19, demonstrating approximately 95% preventive efficacy.
The final inspection committee stated, "The vaccine's safety is generally good. However, close monitoring is necessary after approval for individuals with a history of hypersensitivity including anaphylaxis. Additionally, any adverse events reported in the future will be reflected in the approval details."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














