 Basic Plan for Advanced Regenerative Medicine and Advanced Biopharmaceuticals (Provided by the Ministry of Health and Welfare)
Basic Plan for Advanced Regenerative Medicine and Advanced Biopharmaceuticals (Provided by the Ministry of Health and Welfare)
[Asia Economy Reporter Lee Chun-hee] On the afternoon of the 21st, the government held the "1st Advanced Regenerative Medicine and Advanced Biopharmaceuticals Policy Deliberation Committee" chaired by Kwon Deok-cheol, Minister of Health and Welfare, and announced on the 21st that it reviewed and approved the "Basic Plan for Advanced Regenerative Medicine and Advanced Biopharmaceuticals," which includes the outlook and promotion strategies for the next five years for the development of the advanced regenerative medicine field.
Advanced regenerative medicine is a next-generation medical technology that transplants living cells, etc., into humans to replace or regenerate damaged human tissues and treat diseases. Representative examples include ▲cell therapy ▲gene therapy ▲tissue engineering therapy. It also includes cases using advanced biopharmaceuticals containing human cells, etc., approved by the Ministry of Food and Drug Safety. It can be a new treatment alternative for patients with rare and intractable diseases for which there is currently no treatment, and if successful, the effect can last with just one treatment, making it a core technology of future medicine.
However, due to the nature of regenerative medicine using living cells as raw materials, it has been pointed out that it is difficult to classify products and verify safety under the current pharmaceutical approval system, and there has been a need to establish a new classification and management system.
Currently, Korea possesses considerable technological capabilities, with adult stem cell-based treatment technology at about 85% of the level of the United States, and 44% of stem cell-based therapies approved worldwide being domestic products. However, since 2015, there have been limitations such as minimal visible productization achievements and sluggish research and development outside of stem cells.
Accordingly, the "Act on Safety and Support for Advanced Regenerative Medicine and Advanced Biopharmaceuticals" was enacted and implemented in August last year, establishing a legal foundation, and the first five-year basic plan for K-Regenerative Medicine was formulated. Through this, the government plans to secure global-level advanced regenerative medical technology development and industrial competitiveness based on a trusted advanced regenerative bio safety management system, aiming to become a leading country in Asia.
The government presented three major strategies through this plan: ▲institutionalization of advanced regenerative bio safety management ▲expansion of clinical research and treatment accessibility in advanced regenerative medicine ▲establishment of an innovation ecosystem to promote technology.
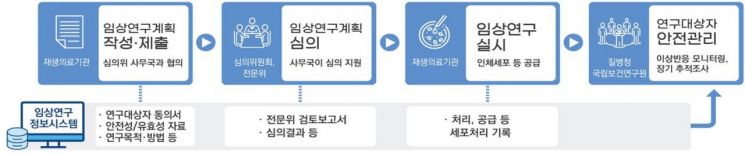 Advanced Regenerative Medicine Clinical Research Management System (Provided by the Ministry of Health and Welfare)
Advanced Regenerative Medicine Clinical Research Management System (Provided by the Ministry of Health and Welfare)
For the safety management institutionalization strategy, a national-level integrated review and management system will first be established for all advanced regenerative medicine clinical research. A central review committee will be formed to examine the appropriateness of research plans, and a clinical research information system will be built to systematically collect research execution data and record and manage the entire process from human cell collection to supply. Safety management of research subjects will also be conducted through this system, and if necessary during or after research, long-term follow-up investigations on the health status, treatment efficacy, and adverse reactions of research subjects will be conducted centered on the Advanced Regenerative Medicine Safety Management Agency (National Institute of Health) after review.
A specialized review and management system will also be established. Specialized industries such as advanced biopharmaceutical manufacturing and human cell management will be newly created, and differentiated quality control systems will be built. By expanding approval and review personnel and introducing a joint review system modeled after advanced countries, a professional approval and review system meeting international standards will be established.
Policy support systems for technological advancement will also be strengthened concurrently. Market entry regulatory system research will be proactively conducted targeting promising technology groups, and especially for technologies with high market entry potential, plans for health insurance coverage will be prepared to ensure no disruption in treatment accessibility. Additionally, a one-stop integrated window will be established to receive corporate difficulties in one place, providing specialized consulting services to promote commercialization of domestic technologies, including R&D investment, overseas regulatory information guidance, and private investment attraction.
For the second strategy of expanding clinical research and treatment accessibility, regenerative medical institutions will be gradually expanded with a target of 200 institutions by 2025, and the annual clinical research budget support, currently about 12 billion KRW, will be more than doubled after 2023 and made eligible for health insurance support as public interest research.
Furthermore, treatment accessibility will be gradually expanded for technologies proven safe and effective through clinical research. When possible to enter medical procedures using the innovative medical technology system, rapid entry will be supported, and limited procedure permissions will also be considered. Separate funding plans will be discussed in addition to expanding national budget support such as patient funds and public foundations.
Alongside this, a public information portal will be established for accurate information delivery, and research on legal, ethical, and social impacts will be conducted to create a public discourse mechanism on related ethical issues and seek solutions, thereby enhancing social trust. Guidelines for illegal procedures will also be prepared and management strengthened.
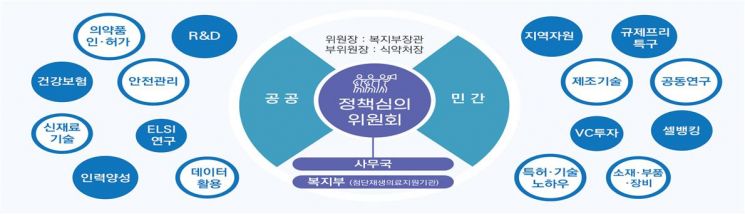 Establishment of Public-Private Integrated Governance for Advanced Regenerative Bio (Provided by the Ministry of Health and Welfare)
Establishment of Public-Private Integrated Governance for Advanced Regenerative Bio (Provided by the Ministry of Health and Welfare)
For the final strategy of establishing an innovation ecosystem to promote technology, a one-stop regulatory and support system will be established. The Advanced Regenerative Bio Policy Committee will serve as the control tower to continuously manage the implementation performance of policy tasks under the jurisdiction of each ministry, and domestic and international networks will be activated. A working group among industry, academia, and government will also be formed for rapid regulatory improvement discussions. The designation of a regenerative medicine regulatory free zone will be pursued, and institutional and technical support measures will be prepared to enable research activation using data accumulated in the clinical research information system.
In addition, national R&D investment will be significantly increased. Approximately 600 billion KRW in national R&D investment will be promoted over 10 years to support technological development throughout the entire cycle of advanced regenerative medicine. Separate support projects will be promoted for self-sufficiency in general-purpose materials, parts, and equipment necessary for advanced regenerative bio R&D, and support will be further increased through the expansion of R&D support targets for small and medium-sized enterprises in this field by the Ministry of SMEs and Startups.
Stepwise expansion of manufacturing infrastructure will also proceed. Technology development projects for production technology innovation, such as general-purpose production and standard process efficiency to reduce manufacturing costs for individual companies, will be promoted. For facilities and equipment with high investment risks, public cell banking services will be provided by expanding infrastructure at the National Stem Cell Regenerative Center and Osong Advanced Medical Complex, and expansion of integrated platform functions supporting cell and gene therapy development will be pursued. Continuous support for professional workforce training systems will also be provided to produce human resources capable of responding to the growth speed of the global bio market.
Kwon Deok-cheol, Minister of Health and Welfare, said, "The ultimate goal of the five-year plan is to develop new treatment technologies to provide better treatment methods to citizens suffering from rare and intractable diseases," and added, "Based on the 1st basic plan, we will actively support turning expectations for advanced regenerative medicine into reality through active cooperation with related ministries, including the Ministry of Food and Drug Safety."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














