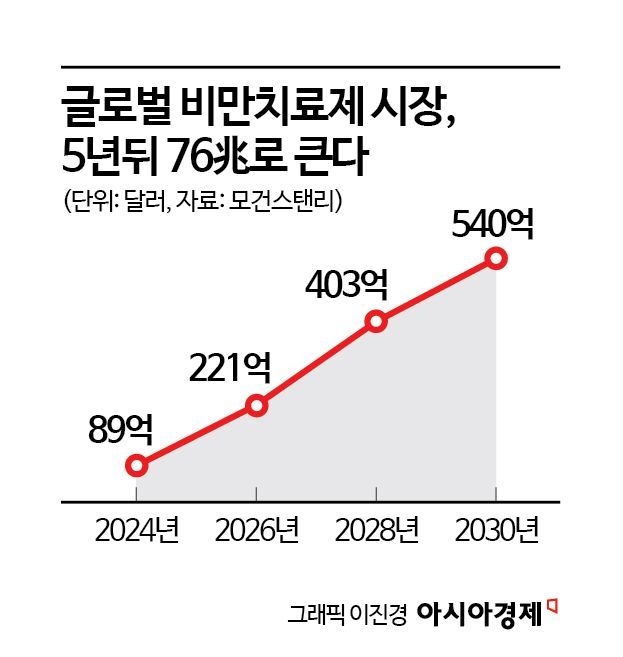
Market Expected to Expand to 76 Trillion Won in Five Years
Competition Intensifies as Oral Obesity Drugs Near FDA Approval
Oral Medications Seen as a Step Closer to "Conquering Obesity"
Industry Anticipates Lower Barriers and Increased Market Growth
The market for oral obesity medications is opening up. Compared to existing products that required abdominal injections, these new oral drugs are considered a step closer to "conquering obesity" due to their ease of administration.
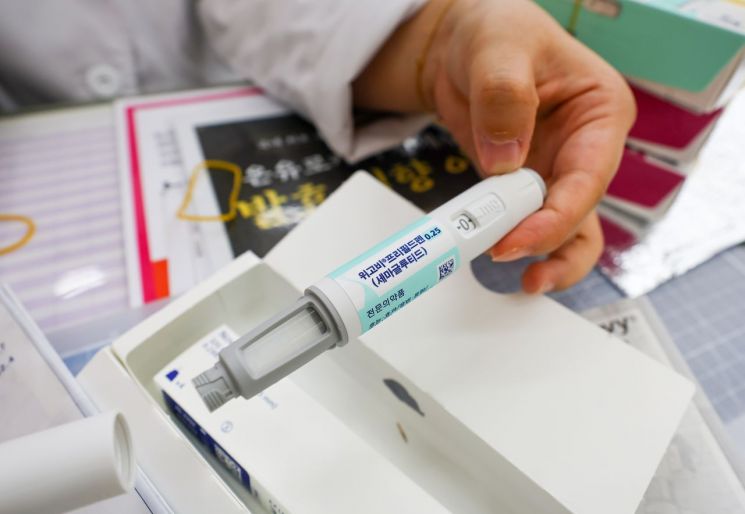 A pharmacist is explaining the obesity treatment drug Wegovy at Saejongno Pharmacy in Jongno-gu, Seoul. Photo by Yonhap News
A pharmacist is explaining the obesity treatment drug Wegovy at Saejongno Pharmacy in Jongno-gu, Seoul. Photo by Yonhap News
According to the industry on April 23, Novo Nordisk and Eli Lilly are each set to apply for FDA approval of their oral obesity treatments this year. It is expected that two oral obesity medications will receive FDA approval and be launched next year.
Novo Nordisk, known for its obesity treatment drug Wegovy, applied for sales approval for the oral version of Wegovy earlier this year. Novo Nordisk reportedly filed for FDA approval based on its successful Phase 3 clinical trial results for oral Wegovy in 2023. In last year's Phase 3 trial, patients who received the highest dose of oral Wegovy lost about 15% of their body weight after 64 weeks. However, Novo Nordisk did not immediately apply for approval, instead focusing on developing next-generation drugs to replace the injectable versions of Wegovy and the diabetes medication Ozempic. The recent application for approval comes as competition with Eli Lilly in the obesity treatment market intensifies.
Eli Lilly recently confirmed significant efficacy in a Phase 3 clinical trial of its new obesity treatment drug Orforglipron. The trial targeted adult patients with type 2 diabetes whose blood sugar was not adequately controlled with diet and exercise. After 40 weeks of treatment, the highest dosage group achieved an average weight loss of 7.9%, and more than 65% of all participants reduced their hemoglobin A1C levels to below the diabetes diagnostic threshold of 6.5%. Both Novo Nordisk's and Eli Lilly's new oral obesity drugs target GLP-1 (glucagon-like peptide-1). GLP-1 is a hormone secreted in the gut after eating, which stimulates insulin secretion crucial for blood sugar control and helps suppress appetite. This mechanism reduces hunger and leads to weight loss.
Developing oral medications is more challenging than injectables. Oral drugs generally have lower absorption rates and may be less effective compared to injectable formulations. The industry has noted that adjusting dosages and establishing delivery systems to achieve efficacy comparable to injectables is complex. Failure to properly adjust dosages can result in side effects such as liver toxicity. Pfizer discontinued development of its oral GLP-1 agonist Danuglipron after observing liver toxicity side effects in clinical trials.
An industry official stated, "The development of oral products lowers the treatment barrier for patients and could significantly expand the obesity treatment market. While competition currently centers on Eli Lilly and Novo Nordisk, if new drugs under clinical development enter the market, treatment prices are also expected to decrease."
According to global investment firm Morgan Stanley, the obesity treatment market is projected to grow rapidly from $8.9 billion (about 12.7492 trillion won) in 2024 to $54 billion (about 77.355 trillion won) by 2030. As the medical costs associated with obesity continue to rise, the UK has recommended insurance coverage for Wegovy for patients with a BMI (body mass index) of 40 or higher. If insurance coverage for obesity drugs expands to other major countries, the global obesity treatment market is expected to grow even further.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














