Maximizing Quantum Sieving Efficiency with High-Entropy 'Cocktail' MOF
Enabling Deuterium Separation at Higher Temperatures
A new porous material has been developed that can efficiently separate deuterium, the fuel for nuclear fusion often referred to as the "artificial sun."
The research team led by Professors Choi Wonyoung and Oh Hyuncheol from the Department of Chemistry at UNIST has developed a metal-organic framework (MOF) capable of separating hydrogen from its isotope, deuterium.
 Research team (from bottom left, counterclockwise) Professor Oh Hyuncheol, Researcher Nam Juhan (first author), Professor Choi Wonyoung, Jung Sungyeop, Jo Changhyun, Researcher Jung Minji. Provided by UNIST
Research team (from bottom left, counterclockwise) Professor Oh Hyuncheol, Researcher Nam Juhan (first author), Professor Choi Wonyoung, Jung Sungyeop, Jo Changhyun, Researcher Jung Minji. Provided by UNIST
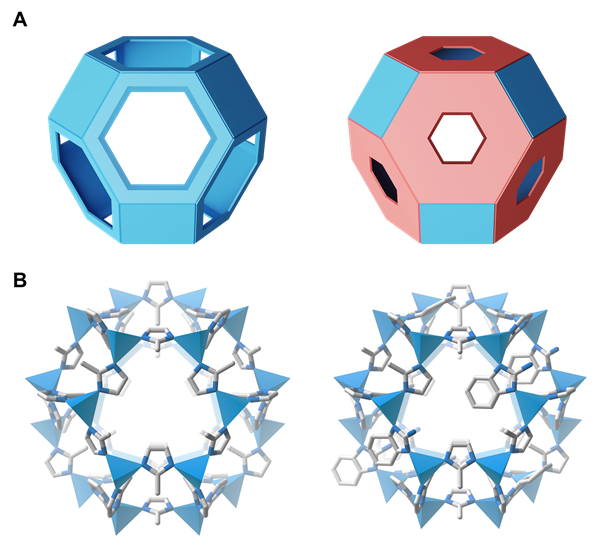
Metal-organic frameworks are structures in which metal ions and organic ligands are chemically bonded to form internal pores. These pores act like a sieve that separates millet from rice, selectively isolating deuterium.
The developed metal-organic framework can separate deuterium from hydrogen even at relatively high temperatures, such as the liquefaction temperature of LNG (111K, -162.15°C).
Typically, deuterium separation is performed at extremely low temperatures below 20K (-253.15°C).
This newly developed metal-organic framework utilizes an entropy-based structural design strategy. By mixing various organic ligands like a "cocktail," the entropy, or degree of disorder, is increased. In this high-entropy state, the quantum sieving effect that separates hydrogen and deuterium is maximized.
Quantum sieving is a technique that separates hydrogen and deuterium by exploiting the difference in their diffusion rates as they pass through narrow pores. As entropy increases, the proportion of narrow pores within the framework also rises, thereby enhancing the efficiency of quantum sieving. The research team confirmed these findings through X-ray diffraction analysis and dynamic breakthrough experiments with hydrogen isotopes.
Professor Choi Wonyoung stated, "This study is the first case of applying high-entropy porous materials to gas adsorption and separation, confirming the potential of entropy-based design," and added, "We hope this technology will contribute to the utilization of clean resources and the advancement of future energy technologies."
Nam Juhan, a researcher from Professor Choi Wonyoung's team, participated as the first author in this study, with researchers Jo Changhyun, Kim Youngjin, Hong Yejin, and Lee Sohyun from Professor Choi's team, as well as Jung Sungyeop and Jung Minji from Professor Oh Hyuncheol's team, participating as co-authors.
The results of this research were published online on December 12 in Angewandte Chemie International Edition, a prestigious journal in the field of chemistry, and are awaiting official publication. The research was supported by the National Research Foundation of Korea (NRF), the Institute of Information & Communications Technology Planning & Evaluation (IITP), and Ulsan National Institute of Science and Technology (UNIST).
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














