Geonjeongsim Excludes 3 Listed Drug Ingredients from Reimbursement Eligibility
The government has decided to increase the health insurance fees for blood products, including whole blood and component blood collection products, and fractionated products, by up to 5,490 KRW. As the medical service gap crisis that began in February continues into its ninth month, health insurance finances amounting to 208.5 billion KRW per month will continue to be injected.
On the 25th, the Ministry of Health and Welfare held the '21st Health Insurance Policy Deliberation Committee Meeting of 2024' and announced the resolution of ▲blood fee increase measures ▲the revision of the 'Drug Benefit List and Ceiling Price Table' reflecting the results of the 2024 drug benefit appropriateness re-evaluation ▲and the extension of health insurance support measures for emergency medical care.
Since 2009, the relative value scores (which represent the relative value of each item to compare the value of each medical benefit item) for blood fees have been fixed, failing to sufficiently reflect the increased costs required for the manufacturing and management of blood products. Accordingly, from January 1 next year, the fees for 39 blood products will be increased by 2,070 to 5,490 KRW per product, reflecting costs such as the unexpected antibody test fees to prevent transfusion side effects and blood collection fees for personnel (230 nurses) involved in the expanded blood management tasks compared to the past.
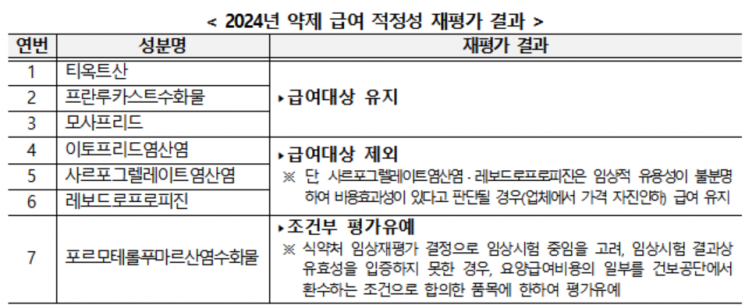
The Ministry of Health and Welfare also conducts an annual re-evaluation of the appropriateness of benefits for drugs listed under the '1st and 2nd National Health Insurance Comprehensive Plans' that require clinical usefulness review due to their long listing period or social concerns.
Among the seven ingredients re-evaluated this year, three ingredients including Thioctic Acid, which were confirmed to have clinical usefulness, will maintain their benefits, while three ingredients including Itopride Hydrochloride, which were evaluated as lacking clinical usefulness, will be excluded from coverage starting November 1. The ingredient Formoterol Fumarate Hydrate, currently undergoing clinical re-evaluation procedures by the Ministry of Food and Drug Safety, has had its evaluation deferred on the condition that if clinical trial results fail to prove efficacy, part of the medical benefit costs will be reclaimed.
The Health Insurance Policy Deliberation Committee also resolved to continue extending the health insurance support for the emergency medical system, which has been in effect since February 20 to prevent treatment gaps for severe and emergency patients caused by doctors' collective actions and to minimize patient inconvenience, at a scale of 208.5 billion KRW per month until the emergency medical crisis phase is lifted.
A Ministry of Health and Welfare official stated, "This resolution follows the '2nd National Health Insurance Comprehensive Plan' announced last February to ensure the stable supply of essential medicines and to adjust the coverage status of items with poor clinical usefulness or cost-effectiveness through re-evaluation. We will contribute to the safe and stable supply of blood products and the sustainability of health insurance."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















