A technology that lowers the depolymerization temperature to improve the efficiency of recycling waste plastics has been developed domestically. Depolymerization is a process that breaks down polymer chains to the monomer level, attracting attention because it allows waste plastics to be chemically decomposed and re-fused. During the depolymerization process, environmentally harmful substances can be filtered out, enabling the production of eco-friendly containers. However, the existing depolymerization process required high temperatures, reducing recycling efficiency. This is where the significance of the developed technology lies.
KAIST announced on the 24th that a research team led by Professor Seo Myeong-eun from the Department of Chemistry developed a method to lower the depolymerization temperature of polymers by utilizing polymer self-assembly.
 The signature is Professor. Provided by KAIST
The signature is Professor. Provided by KAIST
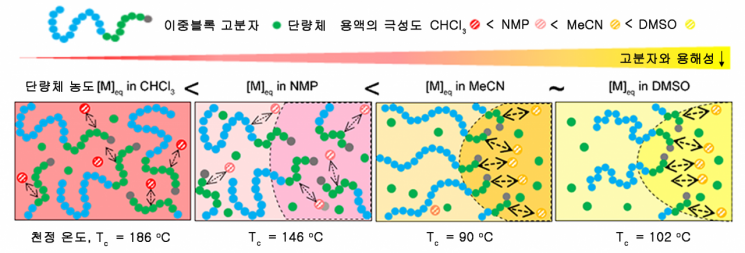
In the conventional process of chemically decomposing polymers through depolymerization, the ceiling temperature (the temperature at which the rates of polymerization and depolymerization are balanced) was 186°C, showing low efficiency in recycling waste plastics.
Accordingly, the research team found a way to induce depolymerization at a ceiling temperature of 90°C, providing a clue to solving the efficiency problem.
First, the team discovered that the depolymerization temperature decreases when self-assembly occurs during the polymer synthesis process. Self-assembly occurring in solvents where polymers do not mix well is a process that creates order against entropy (the tendency toward disorder).
The synthesis process, which connects multiple molecular monomers to form a large polymer chain, also increases order, whereas depolymerization, which breaks polymer chains back into original monomers, is a change toward disorder.
Based on this insight, the researchers confirmed that under conditions where self-assembly occurs, depolymerization dominates over polymerization in balancing order and disorder, enabling the induction of depolymerization at about half the conventional temperature.
 Schematic diagram of polymer self-assembly occurring during polymerization in a selective solvent with low solubility for the block copolymer. Provided by KAIST
Schematic diagram of polymer self-assembly occurring during polymerization in a selective solvent with low solubility for the block copolymer. Provided by KAIST
The research team also implemented a sustainable self-assembly system where, after synthesizing polymers and raising the temperature to break down the polymer chains forming the nanostructure into reusable monomers, lowering the temperature causes the decomposed monomers to polymerize again and form the nanostructure.
Since the shape of the nanostructure varies depending on the length of the chains, the team controlled the temperature and observed changes in the structure’s shape at high and low temperatures. Considering that physical properties such as viscosity depend on the ratio of polymers present among monomers, they confirmed that viscosity could be controlled by repeating polymerization and depolymerization.
Professor Seo Myeong-eun explained, “Previously, high temperatures were required to chemically decompose polymers, making the decomposition process difficult. However, our team was able to lower the depolymerization temperature through polymer self-assembly, and by applying this principle, we found the potential to recycle waste plastics more efficiently.”
Meanwhile, this research was conducted with Dr. Nam Ji-yoon from KAIST’s Department of Chemistry as the first author and Yu Chang-su, a combined master's and doctoral student, as a co-author. The research results were also published in the online edition of the international journal Journal of the American Chemical Society on May 8.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














