Next-Generation Gastric Medicine P-CAB to Replace PPI
Innoen's 'K-CAB' and Daewoong's 'Pexuclu' Approved
Expanding Market Domination from Korea to the World
The domestic pharmaceutical industry is leading the world in the development of treatments for gastroesophageal reflux disease (GERD). With the emergence of the third domestically developed new drug, there is growing anticipation for global market dominance. The focus on a new formulation, the potassium-competitive acid blocker (P-CAB), which enhances both efficacy and convenience compared to existing drugs, is being recognized for its success.
On the 24th, the Ministry of Food and Drug Safety approved Jacubo, a treatment for erosive gastroesophageal reflux disease, as the 37th domestically developed new drug. Jacubo is a P-CAB medication developed by Onconic Therapeutics, a subsidiary of Jeil Pharmaceutical. Jacubo, which contains the active ingredient zastaprazan, received new drug approval after clinical trials showed a higher treatment rate compared to esomeprazole, a proton pump inhibitor (PPI) used as a comparator. This drug is scheduled for release within this year.
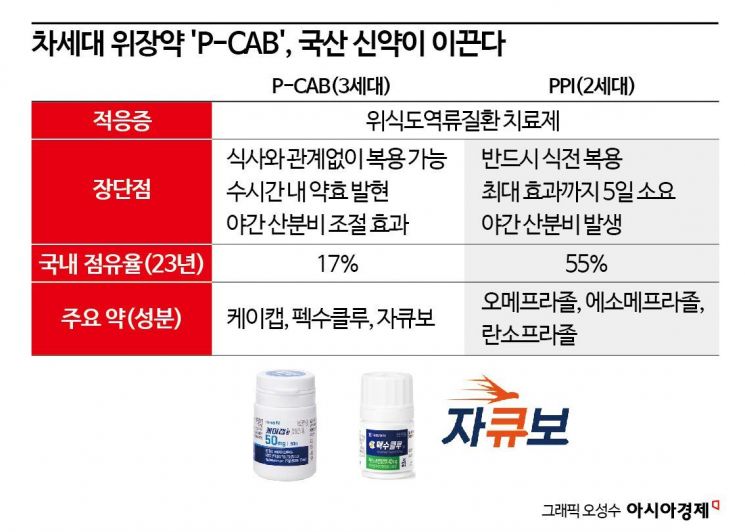
Currently, the most commonly prescribed treatment for GERD is proton pump inhibitors (PPIs), which intervene in the process of gastric acid production. As a second-generation drug, PPIs must be taken before meals because they need to activate before acid secretion, and it can take 3 to 5 days for the effects to manifest. Additionally, the drug is rapidly eliminated from the body, so taking it in the evening can lead to reduced efficacy during sleep, causing heartburn due to acid secretion. P-CABs are classified as third-generation GERD treatments and block acid secretion through a more fundamental mechanism than PPIs. They can be taken at any time regardless of meals, and their effects appear faster and last longer.
Based on these advantages, P-CABs are rapidly capturing the market. According to the pharmaceutical market research firm Ubist, all PPI products among the top 10 prescribed GERD treatments in South Korea saw a decline in sales in the first quarter compared to the same period last year. In contrast, sales of P-CAB products currently on the market, such as HK Inno.N's K-CAB and Daewoong Pharmaceutical's Pexuclu, have grown. The domestic market share of P-CABs rose from 17% at the end of last year to about 20% in the first quarter.
The two leading domestic P-CAB prescriptions, K-CAB and Pexuclu, are accelerating sales growth by expanding their indications. GERD treatments can be used not only for their original indications but also for Helicobacter pylori eradication therapy, treatment of gastric ulcers, and gastritis. K-CAB has secured indications for erosive and non-erosive GERD, Helicobacter pylori eradication, and gastric ulcers. Erosive refers to a condition where the stomach lining is eroded and inflamed. Pexuclu has also expanded its indications to include gastritis. Onconic Therapeutics plans to broaden Jacubo’s indications to include gastric ulcers and other conditions.
Globally, the P-CAB market is expected to grow rapidly. According to global market research firm BCC Research, the P-CAB market in 17 major countries is projected to grow from 61 billion KRW in 2015 to 1.876 trillion KRW by 2030, with an average annual growth rate of 25.7%.
With the approval of Jacubo, three domestically developed new drugs have entered the commercialization stage among P-CAB formulations worldwide. The first global approval of a P-CAB was Takeda Pharmaceutical’s Takecab in Japan in 2015, but since the first domestic approval of K-CAB in 2019, Pexuclu and Jacubo have joined the market. Besides these, only Carephar Pharmaceutical’s Beiwen from China has been approved as a P-CAB new drug in major countries worldwide.
Domestic P-CABs are accelerating their global market entry based on strong domestic prescription performance. K-CAB has already been launched in seven countries across Asia and South America, in addition to South Korea. Including these, it has signed technology or finished product export contracts with a total of 45 countries. Pexuclu is also rapidly expanding its market by obtaining product approvals in the Philippines and three South American countries. Onconic Therapeutics plans to list on the stock market to secure funding for follow-up research based on the development of Jacubo.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)










