UNIST-Stanford International Joint Research Team Develops High-Efficiency Solar Ammonia Production Method
World's Fastest Solar Ammonia Production Rate Recorded … Published in ‘Nature Catalysis’
A technology capable of producing high-efficiency ammonia using only solar energy has been developed.
A research team led by Professors Jiwook Jang and Sungyeon Jang from the Department of Energy and Chemical Engineering at UNIST (President Yong-Hoon Lee), in collaboration with Professor Thomas F. Jaramillo from Stanford University, developed a perovskite-based eco-friendly ammonia production photoelectrode system. The system exceeded the commercialization benchmark for eco-friendly ammonia production rate set by the U.S. Department of Energy by approximately 1.7 times.
This is a world record.
 (Top row from left) First author Ahmad Tayebi, Seongyeon Jang, Jiuk Jang, Professor, (Bottom row) First author Jieun Kim, First author Rashmi Mehrotra, First author Muhibullah Al Mubarak, Dongrak Oh, Researcher.
(Top row from left) First author Ahmad Tayebi, Seongyeon Jang, Jiuk Jang, Professor, (Bottom row) First author Jieun Kim, First author Rashmi Mehrotra, First author Muhibullah Al Mubarak, Dongrak Oh, Researcher.
The principle involves reducing nitrate (NO3-), a major component of wastewater, in water using solar energy to produce ammonia (NH3). The developed system recorded the world’s highest solar-to-ammonia conversion rate and overcame the vulnerability of perovskite to water.
Ammonia, an industrial raw material, is used to synthesize high value-added compounds such as fertilizers, food, and pharmaceuticals. Currently, most ammonia is produced by the Haber-Bosch process, which is not eco-friendly and consumes a massive amount of fossil fuels.
The research team developed a photoelectrode system with high performance and durability by protecting the perovskite solar cell, and a high-performance ammonia production catalyst by depositing ruthenium on titanium nanosheets.
Perovskite absorbs light well and can generate many charges, but it easily decomposes in water. The team protected the perovskite with a fusible metal that easily melts and strongly bonded it with the high-performance ammonia production catalyst. The fusible metal has a low melting point of 63 degrees Celsius, allowing it to melt easily and solidify at room temperature, enabling this design.
The fabricated photoelectrode prevents direct contact between the perovskite and water. It electrically connects and fixes the perovskite and the ammonia production catalyst. When exposed to light underwater, the photoelectrode generates charges. These charges are efficiently transferred to the ammonia production catalyst exposed on the electrode surface, producing ammonia stably and with high efficiency.
To produce ammonia without external voltage, the team used glycerol, which reacts at a lower voltage than water. First, they increased the oxidation reaction rate of glycerol by depositing a platinum catalyst on titanium nanosheets.
The voltage produced by the photoelectrode and the oxidation reaction of glycerol matched the minimum voltage required for ammonia conversion. In other words, when the photoelectrode is immersed in water containing glycerol and exposed to light, ammonia can be spontaneously produced. Simultaneously with ammonia production, glyceric acid, which is more than nine times more valuable than glycerol, was also produced as a byproduct.
The ammonia production rate using the developed photoelectrode reached a maximum of 1745 μgNH3 cm-2h-1. This far exceeds the commercialization benchmark for eco-friendly ammonia production rate of 1000 μgNH3 cm-2h-1 set by the U.S. Department of Energy.
By changing the nitrate reduction catalyst used in the developed system to other types, it can also be applied to the production of various high value-added substances.
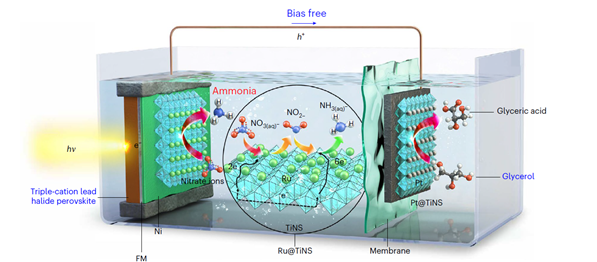 Schematic diagram of an active ammonia production and glycerol oxidation simultaneous process system.
Schematic diagram of an active ammonia production and glycerol oxidation simultaneous process system.
Professor Jiwook Jang said, “Through this research, we produced ammonia and high value-added glyceric acid from nitrate, the main component of wastewater, and glycerol, a byproduct of biodiesel. The developed technology can produce high-efficiency ammonia without external voltage and can also be used to produce various eco-friendly fuels.”
Professor Sungyeon Jang explained, “This research is very important for the application of high-efficiency solar fuel production. It is significant in that it exceeded the solar fuel production rate standard for commercialization.”
The research results were published on April 1 in ‘Nature Catalysis,’ the top journal in catalysis research. Postdoctoral researcher Ahmad Tayyebi, Rashmi Mehrotra, Muhibullah Al Mubarok, and UNIST integrated MS-PhD student Jieun Kim participated as co-first authors.
The research was supported by the Ministry of Science and ICT’s National Research Foundation of Korea through the Excellent Researcher Exchange Support Project (Brainlink) and the Global Basic Research Laboratory Support Project (BRL), among others.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














