The import declaration procedure for medical devices will be simplified.
On the 28th, the Korea Customs Service announced that it will streamline the pre-approval procedure for importing medical devices (Standard Customs Clearance Pre-Notification) in collaboration with the Ministry of Food and Drug Safety (hereinafter MFDS).
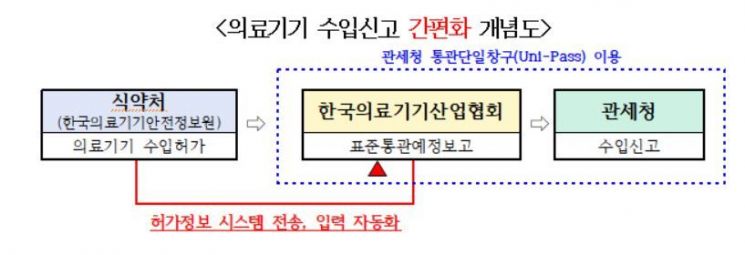
The Standard Customs Clearance Pre-Notification is a procedure that requires importers to obtain approval from relevant associations before import declaration to prevent unqualified or unauthorized importation of medical devices and pharmaceuticals.
Previously, when applicants prepared the Standard Customs Clearance Pre-Notification report through the Korea Customs Service's single window system (Uni-Pass), they had to manually enter the approval information pre-approved by the MFDS and others.
However, going forward, the Korea Customs Service will improve the procedure so that 13 types of pre-approval information received from the MFDS can be automatically entered into the single window system.
The approval information to be automatically entered includes two types of ‘Importer License Information’: the import business license number and the importer license classification, and eleven types of ‘Product Approval Information’: medical device classification number, classification grade code, product name in Korean, product name in English, model name, manufacturer and manufacturing requester’s business name, manufacturer and manufacturing requester’s address, manufacturer and manufacturing requester’s country name in Korean, manufacturer and manufacturing requester’s country name in English, product approval (notification) number, and product approval (notification) date.
The automation of information input is one of the detailed tasks of the ‘Smart Innovation’ initiative that the Korea Customs Service has been focusing on since last year. The Korea Customs Service expects that this automation will contribute to reducing the working hours (33,167 hours) and labor costs (KRW 327.03 million) of medical device importers. It also anticipates preventing errors caused by manual input.
A Korea Customs Service official stated, “In cooperation with the MFDS, we will work to apply information input automation to similar fields such as pharmaceuticals, cosmetics, and human tissues in the future.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














