Medical AI company Lunit announced on the 19th that a research paper predicting the efficacy of immuno-oncology treatment using its AI biomarker platform, Lunit SCOPE IO, was published in the official journal of the Society for Immunotherapy of Cancer (SITC), the Journal for ImmunoTherapy of Cancer (JITC). The "IO" in the platform name stands for Immuno-Oncology.
Founded in 1984, SITC is a global society comprising over 4,600 clinicians, scientists, and researchers from 63 countries specializing in cancer research and treatment worldwide. It is recognized as a world authority in the field of immuno-oncology and serves as an important platform for sharing the latest research findings and innovative therapies.
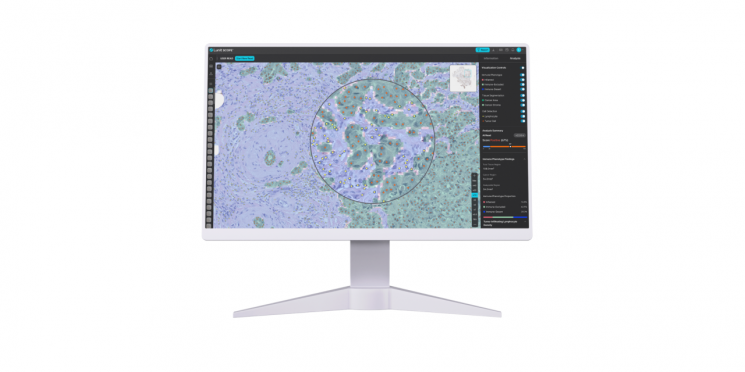
This Lunit study was conducted in collaboration with researchers from five hospitals in Korea and the United States. It analyzed the immuno-oncology treatment response outcomes of 1,806 patients across 27 cancer types. The research team used Lunit SCOPE IO to precisely classify patients' immune phenotypes, which are categorized as immune active, immune excluded, and immune deficient. Among these, immune active patients are generally known to show the most positive response to immuno-oncology treatment.
Analysis results showed that immune active patients exhibited improved metrics in various indicators during immuno-oncology treatment compared to non-immune active patients. The objective response rate (ORR), which is the proportion of patients showing drug efficacy such as tumor size reduction, was 26.3%, higher than the 15.8% observed in non-immune active patients. The median progression-free survival (PFS), indicating the period without further cancer progression, was 5.3 months, and the median overall survival (OS), the total survival time of patients, was 25.3 months?both improvements over the 3.1 months and 13.6 months seen in non-immune active patients, respectively.
Additionally, to comprehensively evaluate the prognostic value of the treatment, several subgroup analyses were conducted considering treatment methods such as immuno-oncology monotherapy, programmed cell death protein (PD)-(L)1 expression rate (TPS), and tissue biopsy sites. As a result, the immune active phenotype classified by Lunit SCOPE was found to be a useful predictor of PFS in most subgroups.
Lunit explained that this study once again demonstrated the clinical significance of predicting immuno-oncology treatment responses using AI biomarkers. In particular, by utilizing Lunit SCOPE, it is possible to logically predict whether the mechanism of action of immuno-oncology drugs fits patients with specific tumor microenvironments, which is expected to greatly contribute to increasing the success rate of clinical trials in the drug development process.
Seobum Seok, CEO of Lunit, stated, “Since immuno-oncology treatment does not have the same effect on all patients, the need for biomarkers to select patients who can achieve good results is gradually increasing. We expect that the results of this study will positively contribute to the design and execution of immuno-oncology clinical trials and ultimately that AI biomarkers will dramatically improve the efficiency of cancer treatment.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















