Government Announces Successive Regulatory Relaxation Measures
Industry Accelerates Business 추진
The domestic direct-to-consumer (DTC) genetic testing industry, which was on the verge of losing momentum due to higher regulatory barriers than overseas, is now breathing new life. This is because the government, which designated the biohealth industry as the second semiconductor industry this year, has been releasing a series of regulatory relaxation measures. The genetic analysis industry is accelerating business promotion by leveraging the regulatory easing trend. DTC genetic testing is a service that allows consumers to learn about their genetic information by requesting private genetic testing institutions without going through medical institutions.
According to related ministries on the 17th, the Ministry of Health and Welfare recently proposed a regulatory relaxation plan for DTC testing as part of the government's joint announcement of the ‘Data Economy Activation Promotion Tasks.’ Representative measures include expanding the scope of permitted tests from wellness items such as hair loss, skin aging, and exercise to ‘disease-like items,’ and simplifying procedures for testing minors. Genetic analysis companies have long complained that various regulations scattered across the country have been blocking the growth of the DTC industry. <Refer to the September 5, 6, and 7 issues of this paper [Stalled Genetic Industry]>
The Ministry of Health and Welfare’s plan to expand the scope of permitted tests to ‘disease-like items’ is interpreted as an attempt to include test items for disease prevention in DTC testing. Genetic testing institutions cannot diagnose specific diseases such as cancer, dementia, or diabetes without consent from medical institutions. A Ministry of Health and Welfare official explained, “For example, it is an attempt to expand the scope to allow testing of genetic material related to blood sugar and the causal relationship between high and low blood sugar, which may be associated with diabetes.” A genetic analysis industry official said, “Although the exact meaning of ‘disease-like items’ needs to be understood, it is unprecedented that the government has used the word ‘disease,’ which was considered a taboo term in the industry.”
The door has opened for adolescents to receive DTC testing. For adolescent testing, valid evidence such as related research papers targeting them is required, but since clinical research targeting adolescents was almost nonexistent, testing for adolescents was practically impossible until now. The Ministry of Health and Welfare plans to simplify procedures so that adolescents can also receive DTC testing by submitting related papers targeting adults and consent forms for adolescents through revisions to the current DTC guidelines. An industry official evaluated, “Because genetic information does not change, there is no difference whether one is 10, 30, or 60 years old. Removing incomprehensible age regulations is a good move by the government.”
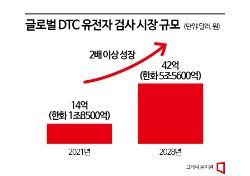
In September, the Ministry of Health and Welfare also expanded the number of DTC testable items from 101 to 129 to enhance the competitiveness of domestic related companies. The government has been implementing the ‘DTC certification system’ since July last year and has been increasing the number of testable items by 20 to 30 each quarter. According to the Korea Bio Association, the domestic DTC testing market size was 30 billion KRW as of last year, and if this trend continues, the market is likely to grow further this year.
Seong Jae-kyung, head of the Bioethics Policy Division at the Ministry of Health and Welfare, emphasized regarding the regulatory relaxation of DTC testing that “it is intended to enhance the competitiveness of the biohealth industry.” Due to the higher level of regulation on DTC testing compared to overseas, domestic companies have had to compete unfairly with foreign companies. Domestic consumers have also turned their attention to overseas companies that offer 3 to 4 times more DTC test items than Korean companies and can predict the degree of disease genetics.
The genetic analysis industry aims to accelerate the expansion of the DTC testing business in line with the government’s continuous regulatory easing trend.
Macrogen, the leading company in the domestic DTC testing industry, welcomed the move, saying, “As regulations are relaxed, the benefits will ultimately return to the public and the nation in terms of improving public health and reducing national medical expenses.” Macrogen launched the DTC testing platform ‘Gentalk’ in June and succeeded in reaching 1 million cumulative users within two months.
Choi Dae-chul, CEO of NGENBIO (Chairman of the Genome Enterprise Council), said, “If the trend of improving the DTC testing system continues, companies will hire people and investments will increase significantly,” adding, “As a result, domestic bio companies will secure global competitiveness, and the industry is expected to be revitalized.” NGENBIO, which recently announced its participation in the U.S. cancer conquest project ‘Cancer Moonshot,’ is conducting unique DTC tests such as muscle strength suitability, morning-type/evening-type personality, and caffeine dependency.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














