HIRA Distributes DTx and AI Guidelines
Leaves Coverage and Non-Coverage Decisions to Companies
Detailed information on the application of National Health Insurance coverage for digital therapeutics (DTx) and artificial intelligence (AI) imaging diagnostic medical devices has been released. Essentially, companies can choose between selective reimbursement and non-reimbursement, with DTx expected to be priced at about 1.5 times the cost, while AI must prove that it offers value beyond existing medical practices to receive separate compensation.
The Health Insurance Review & Assessment Service (HIRA) announced on the 25th that it will distribute revised guidelines for the listing of DTx and AI related to innovative medical technologies under health insurance. This comes about a month after discussions on the 'DTx·AI provisional listing plan' were held at the Health Insurance Policy Deliberation Committee meeting on the 26th of last month, making insurance coverage more tangible.
The newly revised guidelines were prepared through extensive consultations with related academic societies, associations, experts, civic groups, and industry stakeholders to support the rapid clinical utilization of innovative medical technologies that have undergone integrated review and evaluation, thereby generating evidence. The guidelines include ▲ procedures for listing innovative medical technologies under health insurance ▲ application procedures and methods for provisional codes ▲ management of non-reimbursement ▲ and post-management such as monitoring.
Currently, for DTx and AI medical devices, the domestic regulations are regarded as aligning with international standards due to continuous efforts by the Ministry of Food and Drug Safety up to the approval stage. However, concerns have persisted that inadequate regulations at the commercialization stage after approval could hinder industry growth. In fact, even five years after the first approval of AI medical devices, some devices are still used as non-reimbursed in Korea. Most AI medical device companies are expanding overseas where regulations are better established, resulting in a distorted structure where overseas sales account for a larger share of revenue than domestic sales.
First, the policy of allowing companies to choose between non-reimbursement or selective reimbursement (10%) for DTx pricing remains in place. When companies apply for digital medical evaluation, they are given the choice, and if they apply for reimbursement, the Digital Medical Specialized Evaluation Committee evaluates whether the service qualifies for coverage. It is fundamentally stipulated that DTx must be approved by the Ministry of Food and Drug Safety and used through a physician's prescription.
Selective reimbursement is a system where the patient’s co-payment rate is increased to provide coverage when the therapeutic effectiveness or cost-effectiveness is uncertain. Since there are often existing medical technologies that can be substituted and real-world data (RWD) for DTx has not yet been secured, selective reimbursement is seen as a measure to encourage proof of effectiveness. If companies prefer to be evaluated in the market without reimbursement, non-reimbursement is also possible. However, even for non-reimbursed services, the non-reimbursed amounts by healthcare institutions will be disclosed on the HIRA website, and prescription details such as the number of administrations will be required to prevent misuse through monitoring. If actual issues arise, restrictions on non-reimbursement may be considered.
The DTx industry, viewing the sector as still in its early stages, prioritizes inclusion in health insurance to gain public trust and expand its base, showing a higher preference for selective reimbursement. Since it is recognized by the state, it can carry credibility, and patients can expect to reduce some of their cost burden. However, medical institutions may prefer non-reimbursement, so the actual choice between reimbursement and non-reimbursement is expected to involve various factors.
The pricing, which will be composed of medical service fees such as prescription and education (prescription fees), post-use evaluation (effectiveness evaluation fees), and DTx usage fees, was disclosed along with the cost calculation standards on this day. First, the 'unit development cost' concept is applied by dividing the development cost, which includes product development expenses and direct costs, by the expected number of users over three years. Then, profit, maintenance costs, and value-added tax, calculated as 25% of the unit development cost, are added, resulting in a final amount approximately 151?158% of the unit development cost, which is expected to be applied in the future.
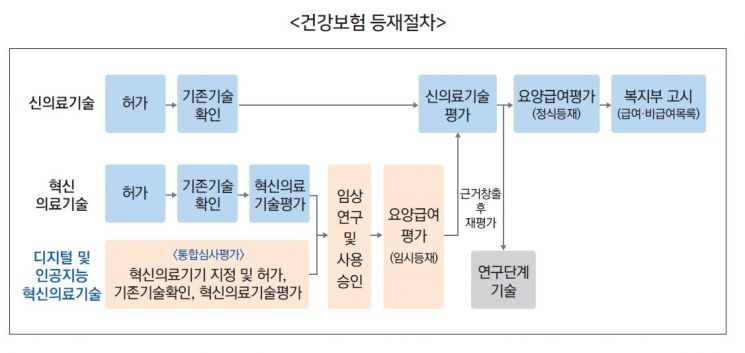 Digital Therapeutics (DTx) and Artificial Intelligence (AI) Technology Health Insurance Listing Procedures
Digital Therapeutics (DTx) and Artificial Intelligence (AI) Technology Health Insurance Listing Procedures Photo by Health Insurance Review & Assessment Service
The reimbursement for medical software applying AI technology is basically similar to DTx but is compensated by adding additional fees (add-ons) to existing fees by categorizing fields into similar categories.
Unlike DTx, specific criteria regarding the nature and performance of the act are presented here. HIRA stated, "If the act is confirmed to be similar to human analysis and interpretation included in existing reimbursed items, and appropriate evidence considering the characteristics of the act proves that accuracy is not inferior to existing acts, it will be recognized as existing reimbursement." Conversely, if it 'replaces existing high-cost medical acts,' 'provides new information beyond the general role of the examination,' or requires additional potential value assessment, it will be recognized as a subject for new medical technology evaluation.
HIRA also announced plans for formal listing in the future. Separate compensation will be granted only if there is a 'significant improvement in diagnostic ability compared to existing acts,' 'creation of new diagnostic value or therapeutic effectiveness,' and additionally, 'proven cost-effectiveness.' In particular, even if AI finds a diagnosis much better than humans, if it does not achieve additional outcomes such as detecting other diseases simultaneously, it is considered difficult to compensate with higher fees or add-ons than the current interpretation fees.
Jung Young-ae, Director of the Reimbursement Listing Office at HIRA, said, "We expect that the guidelines will help easily understand the health insurance listing system for innovative medical technologies and increase predictability for provisional listing." HIRA plans to disclose detailed operational guidelines such as provisional listing calculation standards and claim methods later and will continue to supplement the guidelines through ongoing communication with the field to enable the utilization and evaluation of innovative medical technologies within the health insurance system.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.


![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














