Treatment for Gastroesophageal Reflux Disease
'P-CAB' Formulation Improving on Limitations of Existing PPIs
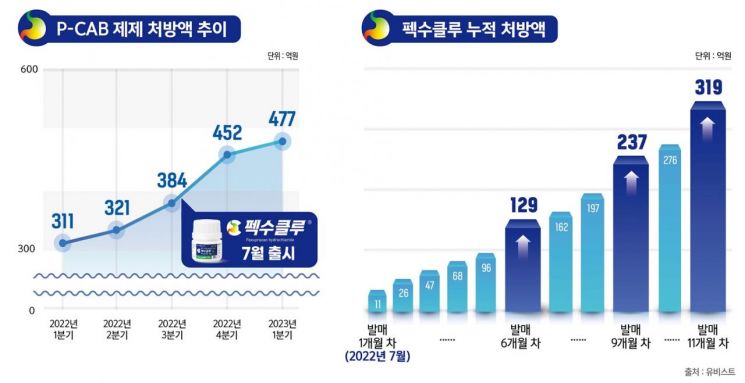
Daewoong Pharmaceutical's potassium-competitive acid blocker (P-CAB) class gastroesophageal reflux disease treatment 'Pexuclu' (active ingredient: Pexuprazan hydrochloride) has surpassed a cumulative prescription amount of 30 billion KRW within 11 months of its launch, driving growth in the P-CAB market.
 Daewoong Pharmaceutical's gastroesophageal reflux disease treatment 'Pexuclu'
Daewoong Pharmaceutical's gastroesophageal reflux disease treatment 'Pexuclu' [Photo by Daewoong Pharmaceutical]
According to UbiStat (UBIST), a pharmaceutical sample statistical information source, as of May, the cumulative prescription amount of Pexuclu is 31.9 billion KRW. Since its launch in July last year, it has maintained an average monthly growth rate of 15%, breaking through 10 billion KRW at 6 months, 20 billion KRW at 9 months, and 30 billion KRW at 11 months, showing an accelerating pace in surpassing milestones.
P-CAB formulations like Pexuclu are considered next-generation treatments that improve upon the drawbacks of existing proton pump inhibitors (PPIs), such as slow onset of action, dietary influences, and drug interactions. In particular, Pexuclu has the longest half-life of 9 hours among gastroesophageal reflux disease treatments, allowing its effects to last long with just a single dose, significantly improving nighttime heartburn symptoms.
Following HK Innoen's 'K-CAB', the addition of Pexuclu has led to continuous growth in the P-CAB treatment market. According to UbiStat statistics, the P-CAB market size in the first quarter reached 47.7 billion KRW, a 54% increase compared to 31.1 billion KRW in the same period last year before Pexuclu's launch.
Daewoong Pharmaceutical plans to accelerate the expansion of Pexuclu's indications and dosage forms for improved convenience. Currently, Pexuclu has secured indications for erosive gastroesophageal reflux disease treatment and improvement of gastric mucosal lesions in acute and chronic gastritis. Additionally, clinical trials are underway to add indications such as maintenance therapy after erosive gastroesophageal reflux disease treatment and combination antibiotic therapy for Helicobacter eradication. Development is also in progress for orally disintegrating tablets that dissolve in the mouth without water and intravenous (IV) formulations to enhance dosing convenience.
Lee Chang-jae, CEO of Daewoong Pharmaceutical, stated, “Surpassing 30 billion KRW in less than a year since launch signifies that Pexuclu has firmly established itself as the next-generation leader among P-CAB treatments.” He added, “Given Pexuclu’s rapid growth, we aim to realize a sales target of around 100 billion KRW within this year and strengthen its position as a ‘game changer’ that shifts the treatment paradigm for gastroesophageal reflux disease from PPI to P-CAB.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














