Next Month's Launch of Biosimilar Market in the US
Coherus Presents 'Yusimli' at Disruptive Price
Market Impact Expected to Be Limited
Low-Concentration Yusimli... Market Already Restructured with High Concentration
Celltrion and Epies Launch High-Concentration Similars
The opening of the US biosimilar (biopharmaceutical) market for Humira (active ingredient adalimumab), worth about 25 trillion won, is just around the corner next month. Including the original, more than 10 products are expected to fiercely compete for market share, and even before the official game begins, intense competition is unfolding, with some companies offering prices as low as 14.4% of the original.
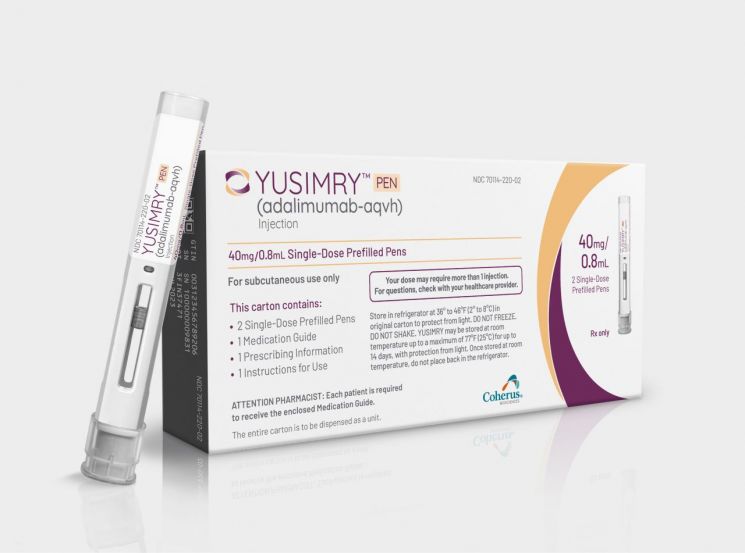
Coherus Biosciences announced on the 1st (local time) that it has priced its Humira biosimilar "Yusimry," scheduled for launch next month, at $995 (about 1.3 million won) for two auto-injectors. Considering that the original Humira costs $6,922 (about 9.05 million won) for two auto-injectors, this represents a price reduction of 85.6%. Accordingly, the annual treatment cost with Yusimry is expected to be about $13,000, significantly lower than the original's $90,000.
This price is much lower than Amgen's "Amjevita," the first Humira biosimilar launched in the US in January, which was priced 55% lower than the original. Since many biosimilar developers who have reached patent agreements with AbbVie, the developer of Humira, are expected to enter the market starting next month, this appears to be an attempt to increase market share through price competitiveness.
Humira, an autoimmune disease treatment, is a global blockbuster with sales reaching $21.237 billion (about 28 trillion won) last year. Although it lost its top spot to Pfizer's COVID-19 vaccine "Comirnaty" during the COVID-19 pandemic, it had maintained the world's top sales position for over a decade prior.
However, with the recent successive launches of biosimilars, its market share has sharply declined in Europe and other regions. In contrast, in the US market, where the biosimilar market has not yet fully formed, sales have actually increased to $18.619 billion (about 24.5566 trillion won), maintaining a growth rate of 7.4% compared to the previous year.
However, industry insiders believe that Coherus's ultra-low price strategy is unlikely to have a significant impact on overall market competition. An industry source said, "Since Coherus has only developed Yusimry as a low-concentration product, it seems to be a desperate measure," adding, "It appears to be a strategy to secure market share even in the low-concentration segment." Given that the US adalimumab market has been reorganized around high-concentration formulations (HCF), Coherus's attempt to target a very small niche market with only low-concentration products is seen as such.
Humira is available in a low-concentration formulation of 50 mg/mL and a high-concentration formulation (HCF) of 100 mg/mL, which reduces the drug dosage by half. While there is still demand for the low-concentration product, preference for HCF is higher. According to pharmaceutical market research firms Symphony Health and IQVIA, 85% of adalimumab sold in the US last year was HCF.
In fact, Amgen's "Amjevita," a low-concentration biosimilar that was the earliest to launch in January and attempted to capture the market, has not exerted significant influence. According to Samsung Securities, Amjevita was prescribed only 662 times in the US in April, with a market share of about 0.3%. This was an increase of only 76 prescriptions compared to 586 in the previous month. Accordingly, Amgen is conducting clinical trials for an HCF version, aiming for market launch next year.
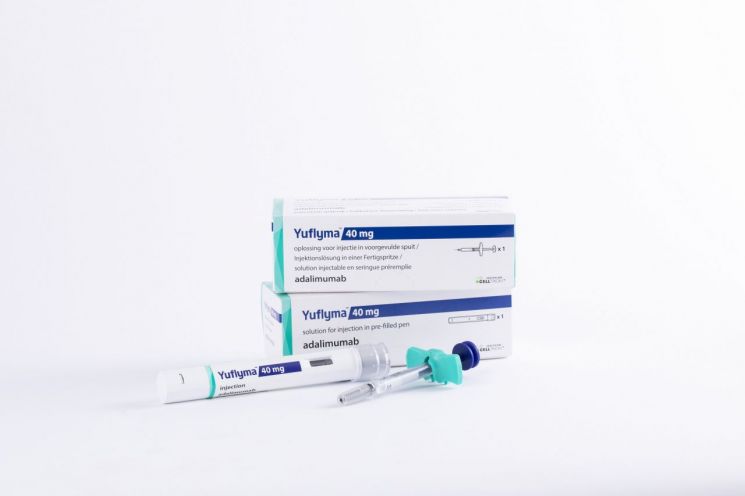
This is why, even when the market opens next month, the real competition is likely to be among HCF products. Currently, there are only four adalimumab HCF products approved in the US: the original, Celltrion's "Yuflyma," Samsung Bioepis's "Hadlima," and Sandoz's "Hyrimoz."
Celltrion developed Yuflyma as an HCF (40 mg/0.4 mL) product that removes citrate, which can cause pain. At the same time, it is conducting global Phase 3 clinical trials in the US and Europe to secure interchangeability with the original, aiming to enhance competitiveness. Sales are directly handled by Celltrion Healthcare, which has recently been expanding direct sales worldwide.
Samsung Bioepis, which succeeded in obtaining FDA approval for Hadlima as the first HCF biosimilar in the US in August last year, also removed citrate to increase market competitiveness. It recently completed clinical trials conducted in four European countries to secure interchangeability. Sales are handled by its US partner, Organon.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














