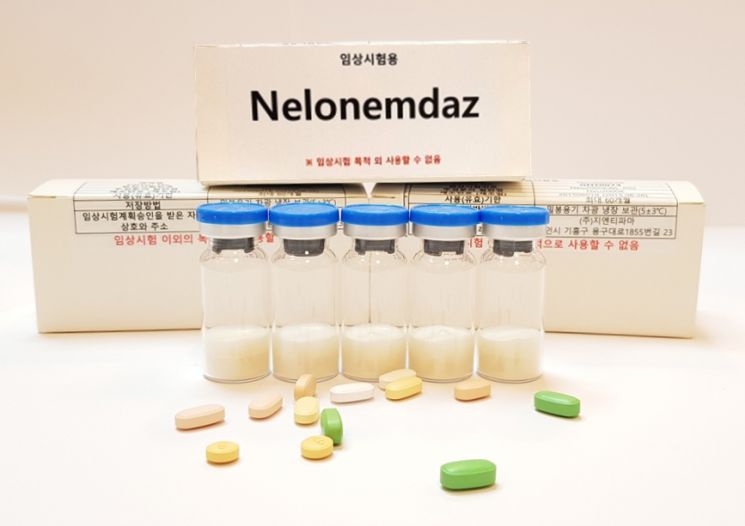
[Asia Economy Reporter Chunhee Lee] GNT Pharma announced on the 26th that it has filed a patent for the lyophilized powder injection of its stroke drug candidate, Nellenemdaz.
This patent covers the manufacturing method, product, and medical use of the lyophilized powder injection of Nellenemdaz. It has been filed in major countries including South Korea, the United States, Europe, and Japan. The lyophilized powder of Nellenemdaz is an injection with excellent moldability, re-dispersibility, high transparency, low impurity content, and improved stability. The patent was filed in key pharmaceutical markets ahead of full-scale production under Good Manufacturing Practice (GMP) standards. Previously, in 2021, patents for Nellenemdaz and its derivatives were filed in the US and internationally (PCT), and it was designated as an orphan drug by the Korean Ministry of Food and Drug Safety.
According to the Global Burden of Disease study report, as of 2019, stroke is one of the top three causes of death and disability worldwide, incurring costs of $891 billion (approximately 1,100 trillion KRW), making it a serious disease. The current standard treatment involves administering thrombolytics within 4 hours and 30 minutes after onset to reopen blocked cerebral blood vessels or performing thrombectomy within 24 hours after onset. However, even after reopening, many patients still suffer from disability and death due to neuronal death caused by glutamate neurotoxicity and reactive oxygen species toxicity.
Nellenemdaz is a multi-target neuroprotective drug that reduces neuronal death after stroke by acting as a safe N-methyl D-aspartate (NMDA) glutamate receptor antagonist and a potent reactive oxygen species scavenger. Currently, a Phase 3 clinical trial is underway at 24 university hospital stroke centers in South Korea, aiming to enroll 496 severe stroke patients who undergo intra-arterial thrombectomy within 12 hours of onset. So far, 396 patients, accounting for 80%, have been enrolled, and the trial is expected to be completed within this year.
Byungjoo Kwak, CEO of GNT Pharma (also an adjunct professor at Yonsei University Department of Life Sciences), stated, "Following the filing of overseas patents for Nellenemdaz for the treatment of stroke patients receiving reperfusion therapy, we have completed domestic and international patent filings for the manufacturing method and use of the lyophilized powder injection, and will actively pursue entry into the global market.” He added, "Depending on the results of the Phase 3 clinical trial of Nellenemdaz, expected to be completed within this year, the stroke drug could be launched as early as the second half of 2024.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














