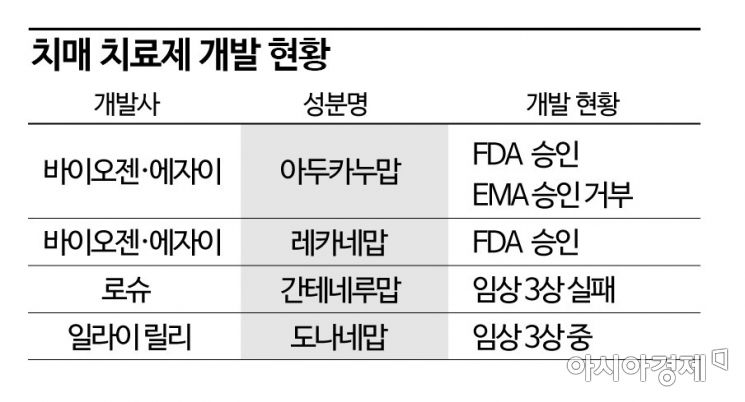
FDA Denies Accelerated Approval for Lilly's Donanemab
Lack of Long-Term Data Beyond 12 Months Cited
Lilly: "Treatment Effects Appeared Quickly, Leading to Early Termination"
Formal Approval Expected After Q2 Phase 3 Completion
Biogen and Eisai's Leqembi Expected to Drive Dementia Treatment Market
[Asia Economy Reporter Lee Chun-hee] Donanemab from Eli Lilly, which aimed to be the third FDA-approved dementia treatment, has faced a setback. However, since the issue is closer to a failure to secure data to prove efficacy rather than an actual problem with the drug's effectiveness, if Donanemab succeeds in entering the market following Biogen and Eisai's 'Leqembi' (generic name: lecanemab), a full-scale competition for dementia treatments is expected to begin.
According to industry sources on the 25th, the FDA recently decided to reject Eli Lilly's accelerated approval application for Donanemab, an Alzheimer's dementia treatment. The FDA reportedly refused approval due to the limited number of patients with at least 12 months of drug exposure. Since there is no long-term follow-up data, the FDA stated it is difficult to verify the related data. Accelerated approval is a system where the FDA grants conditional approval before clinical trials are fully completed for new drugs targeting serious diseases or unmet medical needs.
This rejection is analyzed to be because, in the Phase 2 clinical trial 'TRAILBLAZER-ALZ' submitted by Eli Lilly, a total of 257 patients were recruited, but fewer than 100 patients continued treatment for more than 12 months. This somewhat dampens the heightened expectations following Biogen and Eisai's success in obtaining accelerated approval for 'Leqembi' (generic name: lecanemab) on the 6th (local time), overcoming the setback of 'Aduhelm' (generic name: aducanumab).
However, there is also an assessment that this accelerated approval rejection for Donanemab does not imply concerns about the drug's efficacy. Eli Lilly claims that Donanemab's effect is good, which is why long-term data could not be secured. The clinical trial was designed so that if the level of Aβ plaque removal, which aims to improve symptoms by removing accumulated amyloid-beta (Aβ) plaques, reached a pre-set level, the trial would be stopped. In other words, because the drug was effective, treatment was quickly discontinued, preventing long-term follow-up. Eli Lilly explained that many clinical participants stopped treatment as early as six months due to the rapid plaque reduction.
Accordingly, Eli Lilly is expected to soon complete the ongoing Phase 3 trial 'TRAILBLAZER-ALZ 2' and announce clinical data in the second quarter, aiming for formal approval in the first half of next year. Competitors Biogen and Eisai are also moving quickly. On the day Leqembi received accelerated approval, they submitted a Biologics License Application (BLA) for full approval. They plan to obtain formal approval in the second half of this year and challenge full insurance reimbursement early next year.
 Biogen and Eisai's Alzheimer's dementia treatment 'Leqembi (generic name: lecanemab)' [Image source=AP Yonhap News]
Biogen and Eisai's Alzheimer's dementia treatment 'Leqembi (generic name: lecanemab)' [Image source=AP Yonhap News]
The market outlook is also bright. As the number of dementia patients worldwide rapidly increases, the dementia treatment market is expected to grow rapidly. Pharmaceutical research firm Kotelis forecasts that the Alzheimer's treatment market size will grow from $1.6 billion (about 2 trillion KRW) in 2020 to $5.7 billion in 2030 and $20 billion (about 25 trillion KRW) in 2050, with an average annual growth rate of 29%. With continued development of antibody therapies, the current prescription share of acetylcholinesterase inhibitors at 47% is expected to be dominated by Aβ antibody therapies such as Leqembi and Donanemab, accounting for 69% by 2025.
Global academic information service company Clarivate recently listed Donanemab and Leqembi among 15 new drugs expected to become blockbuster medicines with annual sales exceeding $1 billion by 2027, either launched this year or likely to be approved. Donanemab is projected to generate $1.34 billion (about 1.6536 trillion KRW) in sales, ranking 4th, while Leqembi is expected to earn $1.02 billion (about 1.258 trillion KRW), ranking 8th. However, there remain some question marks about whether these drugs will quickly capture the market. Clarivate noted, "Issues such as insurance pricing, reimbursement, pricing, and early patient identification remain. Since future regulatory and payer decisions regarding Leqembi are likely to set precedents, active use will be difficult until reimbursement conditions are clarified."
Reflecting this situation, Leqembi's price has been significantly lowered. The wholesale price of Leqembi is $26,500 per year (about 32.69 million KRW). Considering that Aduhelm initially set its price at $56,000 but lowered it to $28,200 (about 34.78 million KRW) in January last year and still struggles to enter the insurance market, this price seems to take that into account. Eisai explained, "We estimate the annual value per patient for Leqembi treatment in the U.S. at $37,600," but added, "We set a lower price aiming to expand patient access, reduce financial burden, and support healthcare system sustainability."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














