As Part of a Multinational Clinical Trial
[Asia Economy Reporter Myunghwan Lee] SK Biopharm announced on the 18th that it has submitted a Phase 3 clinical trial plan (IND) for the innovative epilepsy drug "Cenobamate" targeting adolescent generalized seizure epilepsy to the Ministry of Food and Drug Safety.
SK Biopharm's Phase 3 trial will be conducted on domestic adolescent patients with generalized seizure epilepsy. It plans to evaluate the efficacy and safety of Cenobamate in patients aged 12 to under 18 years with primary generalized tonic-clonic seizures.
The domestic Phase 3 trial will involve more than 30 adolescent epilepsy patients, divided into a Cenobamate treatment group and a placebo group, administered for approximately 34 weeks. Afterwards, depending on choice, patients will be able to receive open-label treatment for up to about one year.
Currently, multinational clinical trials are underway to expand the indications for generalized seizures and the eligible age range for Cenobamate (adolescents to adults). As part of these multinational trials, clinical studies targeting adolescents will be conducted in about eight countries including South Korea, the United States, Australia, Germany, and Spain.
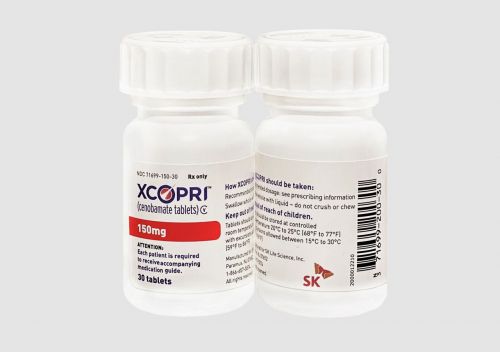
Cenobamate is an innovative epilepsy drug for adults with partial seizures, independently developed by SK Biopharm from start to finish, and was approved for marketing by the U.S. Food and Drug Administration (FDA) in November 2019. Cenobamate has expanded its market across four continents: North America, Europe, Asia, and Latin America. In South Korea, a Phase 3 clinical trial is ongoing targeting adult patients with partial seizure symptoms.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














