5 Global Deals Worth 17 Trillion Won from December to January
Big Pharma Like Amgen, Merck, MSD Engage in Tech Shopping
Domestic Firms Such as Celt, Anguk, Samjin Also Pursue Technology Acquisition
[Asia Economy Reporter Chunhee Lee] What if it were possible to inject poison only into cancer cells like a guided missile to eliminate them? Antibody-drug conjugates (ADCs) that realize this concept have emerged as a hot topic in the pharmaceutical and biotech industries since the beginning of the year.
According to industry sources on the 10th, global big pharma companies are feverishly securing ADC platforms, leading to a series of large-scale ADC technology deals. Since last month alone, a total of five major contracts have been signed, with the combined contract value reaching $13.6725 billion (approximately 17 trillion KRW).
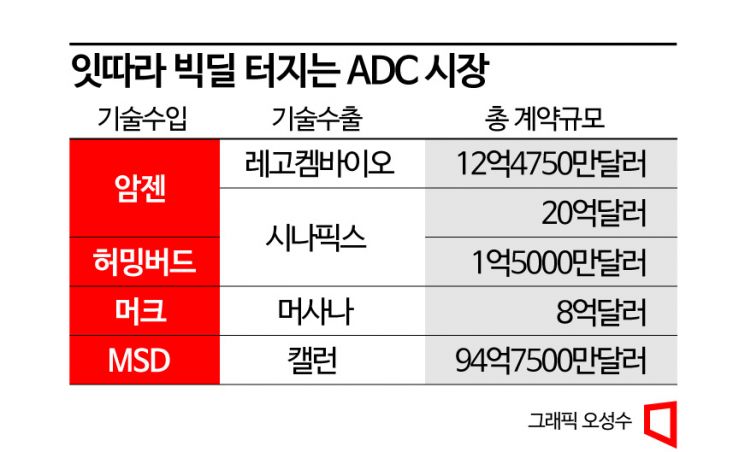
Amgen is the most proactive player. Following a contract last month to purchase ADC core technology from LegoChem Biosciences worth $1.2475 billion (about 1.5331 trillion KRW), Amgen signed an option contract worth $2 billion (about 2.5 trillion KRW) with the Netherlands-based Synaffix on the 5th (local time), intensifying its efforts to secure ADC technology. Amgen has primarily focused on developing antibody drugs and small molecule drugs. Although it had some ADC pipelines, development was halted at an early stage. However, with consecutive technology agreements, it appears to be embarking on full-scale ADC technology development. In addition, Synaffix also signed a $150 million ADC co-development contract with Hummingbird Bioscience, a Singaporean biotech company invested in by SK.
MSD (U.S. Merck) and Germany's Merck are also actively competing to secure ADCs. On the 22nd of last month, MSD signed a massive contract worth $9.475 billion (about 11.8 trillion KRW) with China's Kelun Biotech. The deal includes an upfront payment of $175 million for seven ADC candidates, with milestone payments up to $9.3 billion if Kelun relinquishes all related rights. MSD has signed two ADC contracts with Kelun this year alone and has made another big deal.
Germany's Merck also purchased the ADC platform of U.S.-based Mersana Therapeutics on the 22nd of last month for a total contract value of $800 million, including a $30 million upfront payment. Additionally, it will pay low double-digit royalties separately. Mersana had previously signed an ADC option contract worth $1.46 billion, including a $100 million upfront payment, with GlaxoSmithKline (GSK) in August last year.
The recent surge in interest in ADCs is due to the emergence of drugs that demonstrate truly innovative effects in the ADC field. As the name suggests, ADCs are drugs that conjugate antibodies and drugs. They connect antibodies that bind to cancer antigens with cytotoxic drugs (payloads) that kill cancer cells via linkers, allowing them to deliver toxins effectively only to cancer cells like guided missiles, making them a next-generation cancer treatment technology.
However, if the toxic drugs are not delivered properly, they may affect non-cancerous cells, causing problems that have hindered active drug development until now. For example, the first ADC approved by the U.S. Food and Drug Administration (FDA) in 2000, "Mylotarg," had its approval withdrawn due to liver toxicity issues but was reapproved after reducing the dosage.
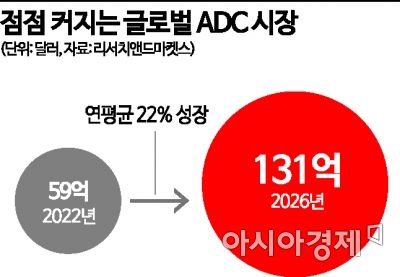
Recently, the ADC "Enhertu," jointly developed by Daiichi Sankyo and AstraZeneca, demonstrated revolutionary results in HER2-low expressing patients, who account for half of metastatic breast cancer patients, receiving standing ovations at the American Society of Clinical Oncology (ASCO) in June last year. Such innovative outcomes have significantly increased market attention. In November last year, ImmunoGen's ADC anticancer drug "Elahere" also received FDA approval, among other new products. Market research firm Research and Markets forecasts that the global ADC drug market will grow from $5.9 billion (about 7.3219 trillion KRW) last year to $13.1 billion (about 16.2517 trillion KRW) by 2026, with an average annual growth rate of 22%.
Accordingly, alliances and collaborations to secure technology are continuing domestically as well. In October, Celltrion secured technology exercise options for 15 targets using the ADC platform from Pinobio. If all options are exercised, the total contract value could increase to a maximum of $1.2428 billion (about 1.5423 trillion KRW). Additionally, Anguk Pharmaceutical signed a joint research contract with Pinobio last month for ADC development, and Samjin Pharmaceutical also signed a joint research agreement with Novelty Nobility on the 4th for ADC development.
Contract development and manufacturing organizations (CDMOs) are also paying close attention to ADCs. Samsung Biologics is reportedly planning to install ADC production facilities at its Songdo Plant 4, where the process is currently being finalized, and Lotte Biologics is considering expanding facilities for ADCs and others at its recently acquired Syracuse plant in the U.S.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















