Blockbuster Drug with Annual Sales of 27 Trillion Won
10 Biosimilars to Launch in the US Next Year
Epics and Celltrion to Enter Domestic Market
Success Depends on Concentration and 'Interchangeability'
Both Epics and Celltrion Secure High-Concentration Formulations
US Senate Moves to Deregulate Interchangeability
[Asia Economy Reporter Lee Chun-hee] The US biosimilar market for the immune-mediated disease treatment drug Humira (active ingredient adalimumab) will open from next year. As it is a mega blockbuster drug market with global sales reaching $20.7 billion (approximately 27 trillion KRW), fierce competition is expected, with Samsung Bioepis and Celltrion from Korea also throwing down the gauntlet.
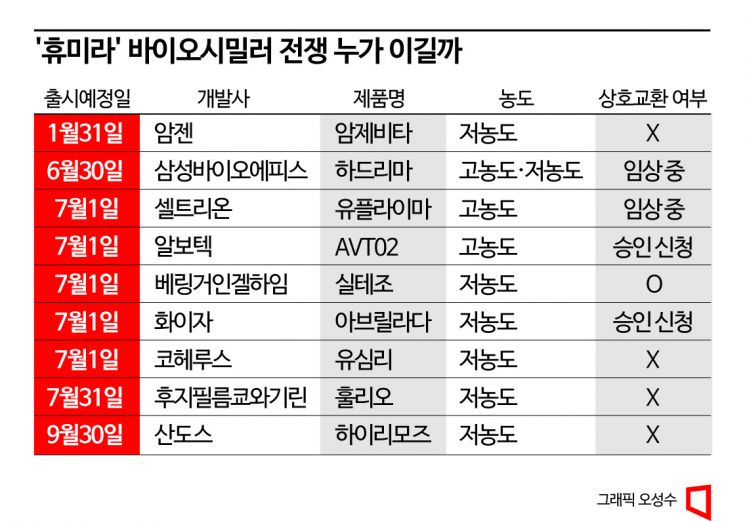
According to industry sources on the 30th, as many as 10 companies have either received or are in the process of obtaining approval from the US Food and Drug Administration (FDA) to launch Humira biosimilars in the US next year. Amgen will launch "Amjevita" on January 31 next year, and Samsung Bioepis will release "Hadlima" on June 30. Competition will intensify in July. On July 1 alone, five products including Celltrion's "Yuflyma," AVT02 (Alvotech), Siltezo (Boehringer Ingelheim), Avrilada (Pfizer), and Yusimri (Coherus) will be launched simultaneously. The launches will continue with Fujifilm Kyowa Kirin's "Hulio" on July 31 and Sandoz's "Hyrimoz" on September 30. Additionally, German Fresenius Kabi is reportedly preparing to launch "Idacio."
Humira, developed by AbbVie, is a treatment for immune-mediated diseases such as rheumatoid arthritis, Crohn's disease, and plaque psoriasis. It is a blockbuster drug that held the world's top sales position for about 10 years. Although it was surpassed last year by Pfizer's COVID-19 vaccine "Comirnaty," which recorded sales of $36.8 billion (approximately 49 trillion KRW), it is widely expected to reclaim the top spot this year.
AbbVie's regional sales are predominantly from the US market, accounting for $17.3 billion (approximately 23 trillion KRW), showing a 7.6% increase compared to the previous year. In contrast, sales outside the US declined by 9.6% to $3.364 billion last year due to the successive launch of biosimilars. AbbVie has tried to block biosimilar entry into the US market through various patent strategies but ultimately agreed with developers to allow biosimilar launches in the US starting next year. AbbVie Chairman Richard Gonzalez mentioned that "Humira sales could be hit by 35-55% starting next year."
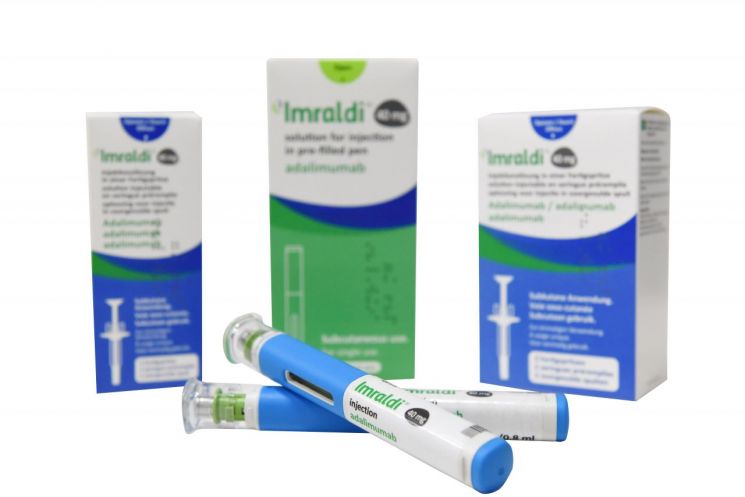 Samsung Bioepis's autoimmune disease treatment 'Humira (active ingredient Adalimumab)' biosimilar 'Imraldi (SB5's European launch name)' (Photo by Samsung Bioepis)
Samsung Bioepis's autoimmune disease treatment 'Humira (active ingredient Adalimumab)' biosimilar 'Imraldi (SB5's European launch name)' (Photo by Samsung Bioepis)
Key factors determining the outcome of next year's Humira biosimilar battle include drug concentration and "interchangeability." These are factors that domestic companies have already prepared for or are seeing a favorable environment for.
Humira is sold in low-concentration formulation (LCF) and high-concentration formulation (HCF). HCF is preferred by patients due to the lower drug dosage. It is known that 80% of Humira sold in the US last year was HCF. Although Amgen's Amjevita is the first to market, it is expected to have limited market impact because only the LCF has been approved so far, with HCF still in Phase 3 clinical trials. In contrast, Samsung Bioepis has developed and received approval for both HCF and LCF for Hadlima, and Celltrion has developed Yuflyma as an HCF-only formulation, giving them a competitive edge.
Securing interchangeability is also crucial. Due to the nature of biologics, which are difficult to produce identically, biosimilars must demonstrate "similarity" rather than equivalence. This is why cross-prescription of biosimilars is not allowed, unlike generics (synthetic drug copies). However, if a biosimilar is deemed interchangeable, pharmacists can substitute it. Currently, only Siltezo has been recognized as interchangeable with Humira. Avrilada and AVT02 have also applied for FDA approval for interchangeability. Hadlima and Yuflyma are conducting clinical trials to secure interchangeability.
Although domestic companies have not yet secured interchangeability, the observation of policy moves abroad that automatically recognize biosimilar interchangeability is seen as a positive development for domestic firms.
Recently, US Republican Senator Mike Lee introduced the "Biosimilar Red Tape Elimination Act." The current system, which requires separate clinical trials and approvals to secure interchangeability, is viewed as bureaucratic "red tape" to be abolished. Senator Lee explained the bill's purpose, stating, "Current regulations make it too difficult and expensive to bring biosimilars to market," and "We will break down barriers that prevent access to drugs that can change consumers' lives." Earlier in September, the European Medicines Agency (EMA) fully allowed automatic substitution for all approved biosimilars, and this trend appears to be strengthening.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














