[Asia Economy Yeongnam Reporting Headquarters Reporter Hwang Dooyul] A catalytic design method that can easily decompose the toxic substance 'anthracene' emitted when using coal, diesel, gasoline, etc., has been developed.
Professor Jaeheung Cho's team from the Department of Chemistry at UNIST recently revealed that the 'manganese-hydroxo species,' one of the metal-active oxygen species, decomposes anthracene, a toxic aromatic hydrocarbon, and that this reaction is based on an electron transfer mechanism.
Anthracene is commonly produced in industrial facilities using fuels such as coal or tar, as well as in exhaust gases from vehicles using fossil fuels.
When mixed in the air, it causes air pollution and is recognized as a carcinogenic substance that induces genetic toxicity and mutations within living organisms, making its decomposition important in the fields of environmental chemistry and biochemistry.
Anthracene is a polycyclic aromatic hydrocarbon composed of three benzene rings, with low solubility and chemical stability.
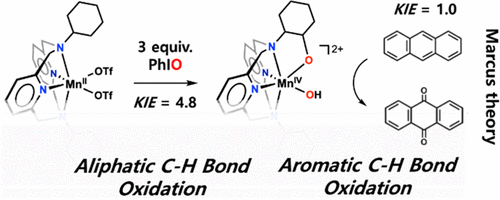
Due to these characteristics, decomposition requires high temperature and pressure, but this study found a way to design a catalyst that can decompose anthracene under low-temperature conditions.
 Dr. Jeong Dong-hyun, Researcher Lee Yoo-jung, and Researcher Son Young-jin, who conducted this study (from left).
Dr. Jeong Dong-hyun, Researcher Lee Yoo-jung, and Researcher Son Young-jin, who conducted this study (from left).
Lee Yujeong, a first author and doctoral researcher in the Department of Chemistry at UNIST, said, “Among aromatic hydrocarbons, ‘naphthalene,’ used as a deodorizer, is very stable, but it is easily decomposed by the natural metal enzyme ‘naphthalene dioxygenase.’ We mimicked this to design a catalytic material that can decompose polycyclic aromatic hydrocarbons, finding a method to decompose them without requiring high temperature and pressure conditions.”
In nature, metal enzymes use external oxygen and electrons to form ‘metal-active oxygen intermediates,’ which directly participate in decomposition reactions.
In this study, ‘manganese-hydroxo species’ was synthesized using iodosobenzene, and the process by which this substance participates in the decomposition reaction of anthracene was revealed. It was found that the ‘manganese-hydroxo species’ decomposes anthracene through an electron transfer reaction.
The analysis of the electron transfer reaction was conducted based on Marcus theory.
Professor Jaeheung Cho said, “Due to the high reduction potential characteristic of the ‘manganese-hydroxo species,’ it decomposes the environmental pollutant anthracene at low temperatures. The academic significance lies in the fact that this is the first analysis of the electron transfer reaction principle causing this phenomenon using Marcus theory. This research will help develop catalysts that decompose environmental pollutants in the environmental and industrial fields in the future.”
Spectroscopic analysis in this study was conducted jointly with Professor J. Roithova from Radboud University in the Netherlands. The research results were published in the prestigious international chemistry journal, the Journal of the American Chemical Society (JACS).
The research was supported by the Basic Research Program (Mid-career Researcher) of the Ministry of Science and ICT and the National Research Foundation of Korea, as well as the Step Leap Carbon Neutral Technology Development Project.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














