Signed Technology Export and Supply Agreement with US Allospecific
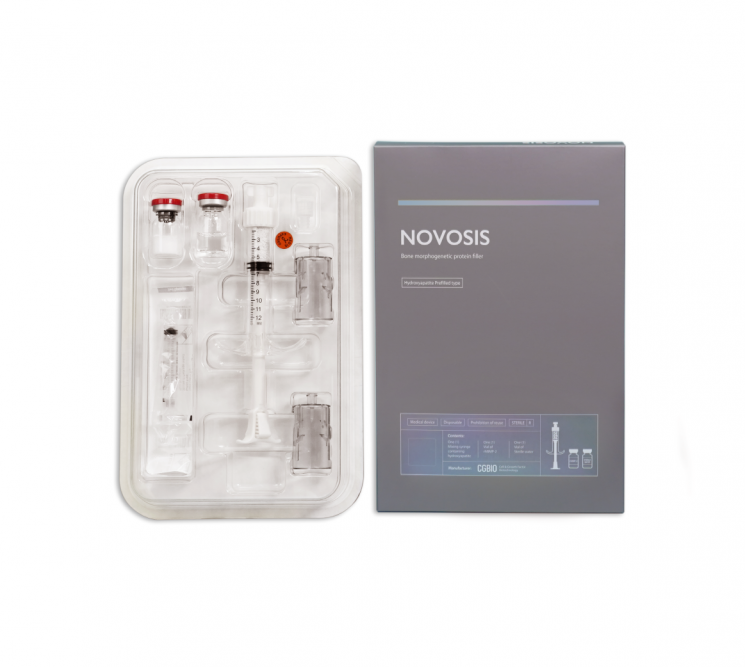
World's Second Successfully Developed Bone Substitute Material
[Asia Economy Reporter Lee Gwan-joo] Sijibio is accelerating its entry into the North American market with its next-generation bone substitute material, 'NOVOSIS.'
On the 5th, Sijibio announced that it had signed a license-out and finished product supply agreement with the U.S. global medical device company Orthofix for the United States and Canada.
The technology fee, including the signing bonus and milestones, amounts to approximately $28 million (about 40.3 billion KRW). The company aims to increase its market share of rhBMP-2-loaded bone substitute materials in North America to 50% over about 10 years from the start of sales after approval by the U.S. Food and Drug Administration (FDA), targeting cumulative sales of around 5 trillion KRW.
NOVOSIS is the first bone substitute material loaded with bone morphogenetic protein developed in Korea and the second in the world. The core technology is a porous hydroxyapatite (HAP) ceramic carrier, which has a structure most similar to human cancellous bone and features excellent biocompatibility. Since bone substitute materials are implanted inside the patient's body and remain for life, biocompatibility is very important. Hydroxyapatite, a material most similar to bone components, is considered the most suitable material for bone substitutes.
Also, since damaged bones recover over several months, it is important that bone morphogenetic protein is released slowly and continuously. Hydroxyapatite allows for the slow release of bone morphogenetic protein, significantly reducing the risk of ectopic bone formation, which is the unintended growth of bone in undesired locations.
The outlook for the bone substitute market is bright. With the global aging trend, the number of patients with disc diseases and senile fractures is increasing. Spinal fusion surgery is essential to treat disc diseases that do not improve with conservative or drug treatments, and in such cases, the use of bone substitute materials is indispensable. According to the global market research firm Smartlock, the North American bone substitute market is expected to reach $2.4 billion (about 3.3 trillion KRW) by 2025.
Meanwhile, Orthofix, which has formed a strategic partnership with Sijibio, was established in 1980 in Verona, Italy, and currently has its headquarters in Lewisville, Texas. It is a global orthopedic medical device company with branches in over 60 countries, conducting distribution and marketing activities for various products.
Yoo Hyun-seung, CEO of Sijibio, said, “This contract is the largest export contract signed by a domestic medical device company and is the result of recognition of Sijibio’s advanced research capabilities and technology.” He added, “It is especially meaningful as it marks the global market entry of Korean bone substitute products. Sijibio will capture the global market with NOVOSIS’s superior technology and competitiveness compared to foreign products.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)










