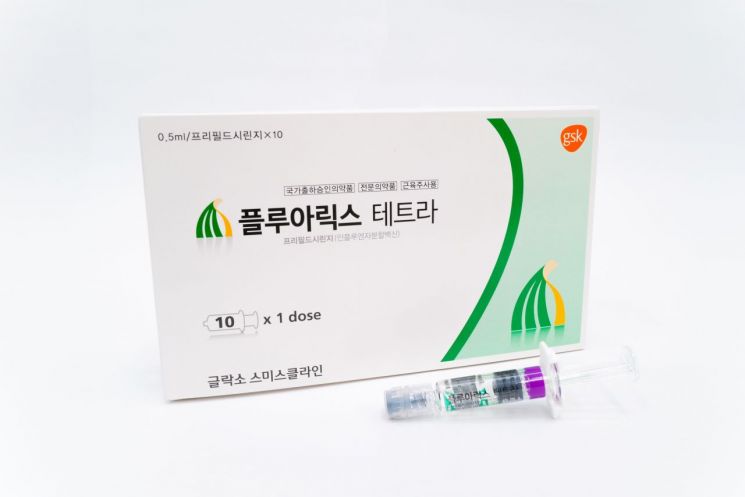
[Asia Economy Reporter Lee Gwan-joo] Kwangdong Pharmaceutical announced on the 10th that it has begun full-scale distribution of GSK's quadrivalent influenza vaccine 'Fluarix Tetra' ahead of this year's flu season.
Kwangdong Pharmaceutical plans to ensure stable and smooth supply of Fluarix Tetra so that vaccinations can be administered nationwide starting in September, the recommended vaccination period.
The typical flu season begins in November, and considering the time required for the vaccine's efficacy to develop, vaccination is recommended from September. This year, Kwangdong Pharmaceutical plans to expand the supply volume to all hospitals and clinics, including pediatrics, obstetrics and gynecology, internal medicine, family medicine, and orthopedics.
Fluarix Tetra is the world's first quadrivalent influenza vaccine approved by the U.S. Food and Drug Administration (FDA). It has demonstrated high safety through extensive usage experience, supplying over 100 million doses in 34 countries worldwide, including Korea.
Kwangdong Pharmaceutical has been co-marketing GSK's pediatric vaccine products since 2015 and has been responsible for the domestic distribution of Fluarix Tetra since last year.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














