[DTx Era⑨] Policy and System in Early Stages
US Leads Global DTx Market
2017: 'Reset' Drug Addiction Treatment
First Approval Worldwide Introduces Concept
Over 10 Approvals Including Insomnia, ADHD
FDA Establishes Regulatory Framework with IMDRF... Proud 10-Year History
Korean Government Late Compared to Advanced Countries
Revising Laws and Providing Support for Development
First Domestic Product Expected This Year
Urgent Need to Address Health Insurance Fees
Approval Must Accelerate to Keep Up with Technology Pace
[Asia Economy Reporter Lee Gwanju] The term digital therapeutics (DTx) has only been widely recognized for less than five years. Before that, terms like digital healthcare, digital medical devices, and digital pharmaceuticals were used interchangeably until the U.S. Food and Drug Administration (FDA) approved Pear Therapeutics' drug addiction DTx 'reSET' in September 2017, marking the emergence of the DTx concept.
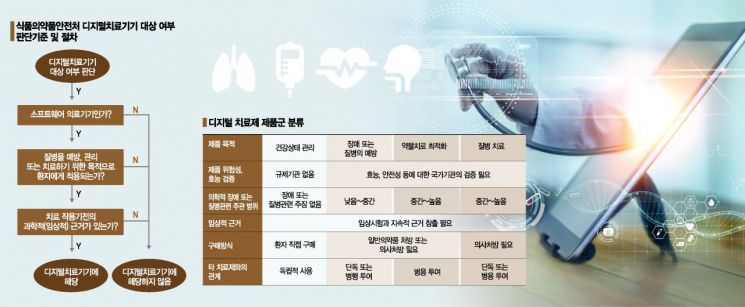
DTx uses IT platforms such as mobile applications (apps) or virtual reality (VR) but is based on medical evidence to provide practical prevention and treatment for specific diseases, distinguishing it from general health management apps. Accordingly, clinical approval and regulatory requirements differ. For example, apps that assist with exercise and provide general health management tips (such as Samsung Health, LG Health) can be developed and marketed without regulatory approval, but DTx must undergo clinical trials and receive formal approval from regulatory authorities, and require a doctor's prescription to be used by patients, similar to conventional drug development processes.
Unlike advanced countries such as the U.S., where DTx has already received formal approval, South Korea is still at the confirmatory clinical trial stage. However, it is evaluated as having global competitiveness in the DTx field as a strong IT and medical powerhouse. Nonetheless, the institutional and policy foundations supporting this are still in the early stages compared to advanced countries that have already established a certain trajectory.
United States Leading Global DTx
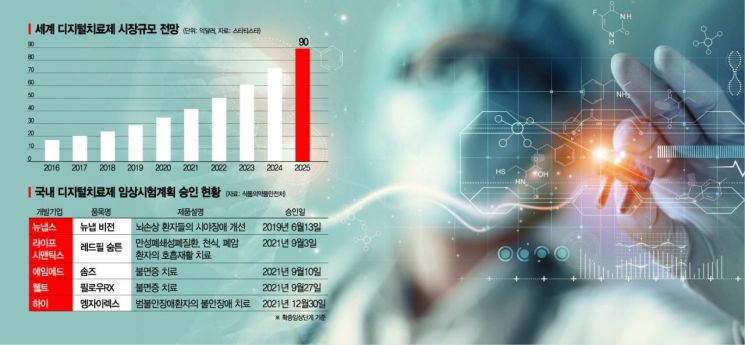
The increase in chronic diseases and the high cost of existing treatments are accelerating the growth of the DTx market. According to the global market research firm Statista, the global DTx market is expected to reach $8.94 billion (approximately 12 trillion KRW) by 2025. The leading country in this market is none other than the United States. Following the world's first approval of the DTx reSET, the U.S. has approved over ten DTx products, including the chronic insomnia treatment 'Somryst' and the pediatric attention deficit hyperactivity disorder (ADHD) DTx 'EndeavorRX.'
The FDA is leading international regulatory policies related to DTx. Since 2013, it has formed a working group with the International Medical Device Regulators Forum (IMDRF) to incorporate and manage DTx as a type of Software as a Medical Device (SaMD), establishing a regulatory framework. This framework boasts nearly a decade of history. Through this, four regulatory guidelines have been established covering the definition of DTx, risk-based classification systems, quality management systems, and clinical evaluation criteria. Notably, recognizing that traditional randomized controlled trials for drugs are ineffective in the rapidly evolving DTx field, a new approval system has been developed.
A representative example is the FDA's 'Pre-Cert Pilot Program' and designation of 'Breakthrough Devices.' Instead of certifying products, qualifications are granted at the developer level, simplifying the approval process by collecting and submitting data post-market rather than pre-market, and allowing updates without requiring medical device change approvals. This is significant because under previous regulations, every update to a DTx would have required separate approval, but these unnecessary regulations have been eliminated. For instance, when Pear Therapeutics first received approval for reSET, it submitted randomized controlled trial results as it was a new treatment method, but its follow-up product 'reSET-O' was designated as a breakthrough device and obtained FDA approval within one year.
South Korea in Early Stages but "Sufficient Competitiveness"
The South Korean government has also begun revising laws and providing support related to DTx development. However, compared to advanced countries like the U.S. and Europe, it is somewhat late. The 'Medical Device Industry Promotion and Innovative Medical Device Support Act' was first enacted in May 2020, and in August of the same year, guidelines for approval and review of digital therapeutic devices were announced. In December last year, the government continued its development-friendly approach by establishing the 'Guidance for GMP Operation of Software Medical Device Manufacturing Facilities.'
Notably, the Ministry of Food and Drug Safety (MFDS) certifies manufacturers of medical device software designated as innovative medical devices and can exempt some documentation required for manufacturing approval and certification. This appears to partially adopt the FDA's related system. Although no DTx has been formally approved yet, five DTx products have entered the confirmatory clinical trial stage, which is the step before formal approval.
Related companies positively evaluate the government's proactive policies. Kwon Hee, director at LifeSemantics, which is conducting confirmatory clinical trials for the respiratory rehabilitation DTx 'Redpill Sumtun,' said, "The MFDS is making efforts to be business-friendly," adding, "We are jointly considering ways to continuously activate the industry." Kang Sangwook, executive director at F&I Korea, developing nicotine and alcohol addiction DTx 'Nicotera' and 'Alcotera,' also said, "It feels like the MFDS is moving very quickly," and evaluated, "The fact that they created a new department (Digital Health Regulatory Support Division) that did not exist before shows they have prepared a lot and means they intend to push forward institutionally."
Voices calling for further institutional improvements still exist. The first issue raised is the health insurance reimbursement fee. Due to the characteristics of the domestic health insurance system, reimbursement fees must be set for actual prescriptions to be made. If this is difficult, non-reimbursed prescriptions should at least be possible. Since the first domestic DTx is expected to be released as early as this year, the industry unanimously calls for swift and clear criteria regarding reimbursement fees.
There are also calls to revise the strict approval system to reflect the characteristics of DTx. Kim Jinwoo, CEO of Hi, which has entered confirmatory clinical trials for the generalized anxiety disorder DTx 'Anzeilex,' pointed out, "DTx includes social and psychological concepts, but the existing perspective that strictly views mechanisms is still reflected," adding, "Although South Korea is a strong IT and bio country, the combined field has yet to stand out globally." He continued, "South Korea has excellent infrastructure such as the 'Big 5' hospitals and is a really good testbed for clinical trials," emphasizing, "If laws and systems are slightly improved, it can definitely become a global hub for DTx."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















