[Asia Economy Sejong=Reporter Kim Hyewon] The medical device software (SW) change approval system will be converted into a negative regulation system. This means that minor changes such as maintenance, repair, and security function updates of medical device software will no longer require change approval.
Also, rental and lease car companies and online shopping businesses will be able to receive separate national subsidies when purchasing electric vehicles in local governments where they have not established branches. The system for posting veterinary hospital treatment costs, which has been controversial due to excessive billing, will be made more specific by item.
On the 10th, the government held the 499th Regulatory Reform Committee meeting at the Government Seoul Office and announced on the 13th that it had discussed and finalized the 'New Industry Corporate Regulatory Improvement Plan.'
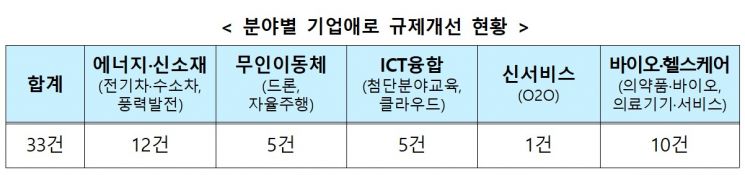
The New Industry Regulatory Innovation Committee, composed of 120 private experts, held 19 meetings and identified 33 regulations to improve in new industry sectors such as electric vehicles and hydrogen vehicles, wind power, drones and autonomous driving, ICT convergence, and bio-healthcare. Among these, three have been completed, and the remaining 30 tasks will be promptly addressed through legal revisions and administrative measures.
An official from the Office for Government Policy Coordination said, "In the context of the Yoon Seok-yeol administration promoting comprehensive regulatory innovation to expand domestic and foreign corporate investment and economic growth, it is meaningful that businesspeople, experts, and public officials have come together to swiftly prepare improvement tasks to resolve unreasonable regulations in new industry fields." He added, "We will continue to strive to resolve regulations that act as obstacles to investment in new industries in the shortest possible time."
A notable example is the conversion of the medical device software change approval system into a negative regulation system. Medical devices require change approval when their approved items are altered, and minor changes as defined by the Ministry of Food and Drug Safety (MFDS) notification are exempted. However, due to the nature of medical device software, maintenance, repair, and security function updates occur very frequently, but these are not included in the minor changes category, resulting in industry burdens such as the time required for change approval (an average of 42 days) and fees (an average of 1 million KRW). Therefore, to reduce the burden on medical device businesses and provide administrative convenience, the government decided to specify 'significant changes' instead of 'minor changes' and require change approval only in those cases, while allowing companies to self-manage changes not falling under significant changes under a negative regulation system.
Additionally, corporations (rental and lease car companies) can now receive separate national subsidies when purchasing electric vehicles in local governments where they have not established branches. Previously, corporations operating electric vehicles nationwide could not receive separate subsidies from each local government due to difficulties in establishing branches. The payment criteria will be finalized through inter-ministerial consultation and included in next year's electric vehicle subsidy project operation guidelines. It is estimated that about 40,000 electric vehicles will receive separate national subsidies.
To enable timely use of the latest drone equipment, special flight safety standards will be more comprehensively defined, and approval will be required after safety inspections by the Aviation Safety Technology Institute. Although special flight approval from the Minister of Land, Infrastructure and Transport is required for drone night flights, the safety equipment and facilities required under the special flight safety standards were specifically stipulated, preventing the use of the latest equipment due to technological advancements.
For approximately 5,000 facilities handling small quantities and with low risk in chemical accident prevention management plans, inspection intervals will be extended. This aims to alleviate operational burdens by differentiating inspection intervals according to hazardous chemical handling facilities.
The installation of sterilization and crushing facilities for medical waste in hospitals will be expanded. Although medical waste generation is rapidly increasing, there are only 14 dedicated incinerators nationwide, resulting in burdens of long-distance incineration costs (about 200 billion KRW annually) and concerns about secondary infections in case of accidents. Accordingly, the Ministry of Health and Welfare has issued an authoritative interpretation that medical waste treatment facilities such as sterilization and crushing facilities are included as ancillary facilities (extraction treatment facilities) of medical institutions. It is expected that installation of sterilization and crushing facilities will become easier in about 140 hospitals nationwide.
The veterinary hospital treatment items and treatment fee standards will also be made more specific. This aims to reduce consumer complaints caused by excessive treatment and overcharging. Although the posting system for veterinary hospitals was introduced through amendments to the Veterinarian Act, specific treatment items and posting methods have not yet been established. Therefore, treatment fees will be posted by item, such as basic examination, hospitalization, vaccination, testing, and interpretation fees.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














