[DTx Era②] Even When Diagnosed, Treatment Hesitated Due to 'Stigma' Concerns
Digital Therapeutics Reduce Burden on Parents and Children
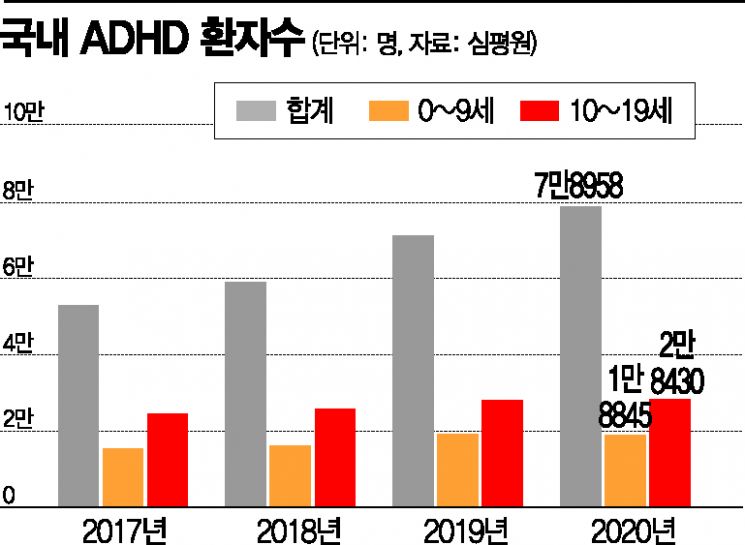
[Asia Economy Reporter Lee Gwan-joo] Attention Deficit Hyperactivity Disorder (ADHD) is generally viewed in the medical community as having many more “hidden patients” who remain undiagnosed than the actual number of confirmed cases. According to the Korean Academy of Child and Adolescent Psychiatry on the 24th, the estimated prevalence of ADHD in South Korea is about 5.9?8.5%, but data from the National Health Insurance Service shows an ADHD diagnosis prevalence of only 0.8%. This means that only about 10% of children with ADHD receive treatment, indicating a significant lack of social awareness.
The global market size related to ADHD treatment is expected to continue growing steadily. The global market research firm Grand View Research projected that the worldwide ADHD market, which was $16.4 billion (approximately 20 trillion KRW) in 2018, will grow to $24.9 billion (approximately 31 trillion KRW) by 2025. However, current ADHD medications are very limited, have serious side effects, and some contain narcotic components, making them unsuitable for prescription to children and adolescents.
Therefore, there is a strong demand for the development of effective ADHD digital therapeutics. Currently, the only ADHD digital therapeutic (DTx) globally recognized is “EndeavorRX,” developed by Akili Interactive, which received approval from the U.S. Food and Drug Administration (FDA) in June 2020.
EndeavorRX treats attention deficits in children aged 8 to 12 through a video game format. It is used for at least four consecutive weeks, five days a week, for 25 minutes each session, and its effectiveness has been confirmed in major clinical trials. In five studies involving 600 participants, 68% of parents reported improvements in ADHD-related symptoms two months after treatment. Additionally, 73% of children showed improved attention, and no serious side effects were observed. EndeavorRX has been commercialized in the U.S. and is preparing for global market entry, including ongoing clinical trials in Japan.
If domestic companies lead the development of ADHD DTx, prove its therapeutic effects, and officially commercialize it, the market value is expected to be very large. Industry insiders believe that more proactive development and support are necessary to enjoy such first-mover advantages. Kim Ho-young, Chief Operating Officer (COO) of Hi, which is developing the ADHD digital therapeutic “ForMe,” said, “Many parents still do not acknowledge ADHD or hesitate to seek treatment due to stigma concerns, but DTx can reduce the burden on these parents and schools. Like in the U.S. and Europe, South Korea also needs to shift its perspective and embrace digital therapeutics.”
Domestic companies are also stepping up their efforts. Hipoti&C’s software “AttnKare,” developed using virtual reality (VR) and artificial intelligence (AI), won innovation awards in two categories?Virtual/Augmented Reality and Digital Health & Wellness?at CES 2022, demonstrating its competitiveness. The software analyzes and evaluates children’s responses while they wear VR devices and complete missions, then provides customized games accordingly. The DTx “COLLITEE,” jointly developed by DarwinTech and NuxBio, also targets ADHD as an indication.
Park Hyuk-tae, head of the Industrial Policy Team at the Korea Creative Content Agency, emphasized, “Since DTx development does not conclude within one or two years, active government support is necessary to sustain research and development. If networking among companies, medical professionals, and other fields essential for DTx development is established, it could lead to more vigorous DTx development.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














