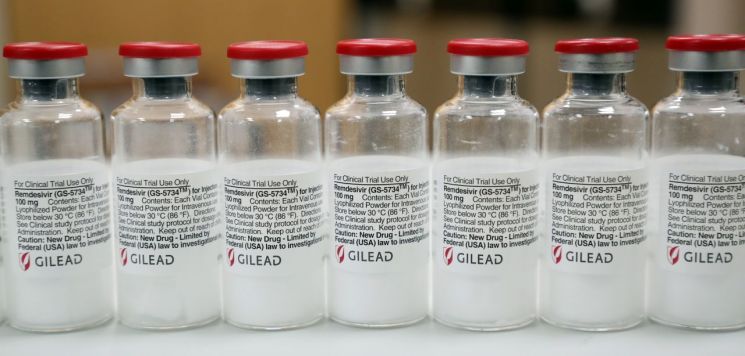
[Asia Economy Reporter Hwang Sumi] Glass particles were found in the COVID-19 treatment injection Remdesivir, prompting the American pharmaceutical company Gilead to recall 55,000 vials.
On the 4th (local time), according to the Washington Post (WP) and others, Gilead recalled enough Remdesivir vials to treat 11,000 hospitalized COVID-19 patients in the United States. These vials have been distributed nationwide in the U.S. since last October.
Gilead stated in a press release that customer complaints reported the presence of glass particles in Remdesivir, and investigations confirmed this fact.
Gilead said, "Administering injections containing glass particles can cause inflammation or swelling and may block the heart, lungs, or cerebral blood vessels, potentially causing stroke and death," but added that no adverse event reports have been received so far.
Gilead also said that they have sufficient stockpiles of COVID-19 treatments, so this recall will not affect the supply of treatments to the U.S. or other countries.
Meanwhile, a similar case of foreign substances found in injections occurred last September in Japan, where stainless steel components were discovered in Moderna's COVID-19 vaccine, leading to the suspension of 1.63 million doses.
At that time, Moderna and Takeda Pharmaceutical, responsible for supplying the Moderna vaccine in Japan, jointly stated, "The most likely cause of contamination is related to the friction between two metal parts of the machine that caps the vials."
In addition, as foreign substances were found in unused vaccines or rubber pieces appeared in vials due to incorrect needle insertion, three regions in Japan temporarily suspended the use of Moderna vaccines with different batch numbers.
Last September in South Korea, an influenza vaccine with white particles was also recalled.
At that time, the Ministry of Food and Drug Safety held a briefing at the Osong Disease Control Headquarters in Chungbuk and announced that Korea Vaccine, the manufacturer of the influenza vaccine, voluntarily recalled 615,000 doses (4 manufacturing lots) of its influenza vaccine 'Kovax Flu Quadrivalent PF Injection' due to the discovery of white particles.
Korea Vaccine recalled two manufacturing lots where white particles were confirmed and proactively recalled two other manufacturing lots that used the same syringes but where white particles were not detected.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














