KBSI and Domestic Research Teams First in the World to Elucidate Carbonation Reaction Mechanism of Calcium Hydroxide
New Clues for Carbon Dioxide Storage and Conversion Technologies for Carbon Neutrality
Published in International Chemical Engineering Journal

[Asia Economy Reporter Kim Bong-su] Domestic researchers have elucidated the principle by which water promotes the reaction between calcium hydroxide, a major component of limestone, and carbon dioxide. This is expected to provide new clues for technologies related to the reduction, conversion, and industrial utilization of carbon dioxide (CO2), a major cause of global warming.
The Korea Basic Science Institute (KBSI) announced on the 8th that Dr. Han Ok-hee's research team at the Seoul Western Center discovered that water plays a crucial role by inserting itself into the layered structure during the early stage of the carbonation reaction of calcium hydroxide, thereby accelerating the carbonation reaction, and successfully measured the amount of inserted water.
Calcium hydroxide, mainly used for detecting carbon dioxide, can convert the greenhouse gas CO2 into calcium carbonate or calcium bicarbonate through carbonation reactions, making it useful in devices that process carbon dioxide. A representative example of the carbonation reaction is the principle by which cement hardens when mixed with water, as calcium oxide in the cement reacts with atmospheric CO2 dissolved in water. Although it is widely known that the carbonation reaction of calcium hydroxide occurs rapidly when mixed with water, the role of water in the reaction process has not been clearly elucidated, nor has the amount of water involved been quantified until now.
Conventionally, it was believed that water adsorbs on the surface of calcium hydroxide to aid the carbonation reaction, but this study revealed through XRD analysis that the interlayer spacing of the layered calcium hydroxide structure expands during the early stage of the carbonation reaction.
The joint research team used solid-state NMR analysis to demonstrate that water molecules enter between the layers of calcium hydroxide at the early stage of carbonation and measured the amount of water inserted. Analysis of calcium hydroxide samples mixed with water showed that while ordinary water exhibits hydrogen isotope signals at 4.8 ppm, water molecules inside calcium hydroxide produced signals around 1 ppm.
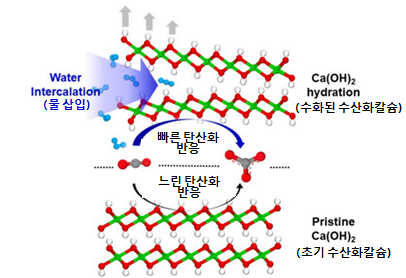 Schematic diagram of the expansion process of calcium hydroxide due to water insertion affecting the carbonation reaction
Schematic diagram of the expansion process of calcium hydroxide due to water insertion affecting the carbonation reaction
By utilizing the distinct signal area sizes, the amount of water inserted between the layers of calcium hydroxide could be quantified. Additionally, theoretical calculations considering reaction rates based on the chemical properties of various substances under specific conditions confirmed that carbonation proceeds faster when water first enters between the layers of calcium hydroxide.
This result is the world's first case to empirically demonstrate the correlation between the structure of water-inserted calcium hydroxide at the atomic level and the carbonation reaction. In the future, calcium hydroxide is expected to be used as a capture agent to reduce factory exhaust gases and atmospheric CO2, as well as in various research and industrial fields such as controlling the hardness of cement and enhancing the properties of fillers by carbonating high-purity calcium hydroxide to improve the functions of paint and toothpaste.
The research findings were recently published in the international chemistry journal 'Chemical Engineering Journal.' The study was conducted in collaboration with Dr. Lee Seung-woo's research team at the Korea Institute of Geoscience and Mineral Resources, Dr. Kim Yong-il's team at the Korea Research Institute of Standards and Science, and Professor Kwak Sang-gyu's team in the Department of Energy Chemical Engineering at Ulsan National Institute of Science and Technology (UNIST).
Dr. Han stated, "This research well demonstrates the advantages of NMR analysis techniques that can simultaneously elucidate the structure and perform quantitative analysis of compounds at the atomic level," adding, "We will continue to strive to contribute to achieving carbon neutrality goals such as renewable energy and CO2 storage and conversion through diverse applications of NMR technology."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














