[Asia Economy Reporter Kim Suhwan] The external advisory committee of the U.S. Food and Drug Administration (FDA) has made a decision to recommend Pfizer vaccine administration for children aged 5 to 11.
According to major foreign media on the 26th (local time), the FDA advisory committee, the Vaccines and Related Biological Products Advisory Committee (VRBPAC), held a meeting related to COVID-19 vaccines and voted accordingly.
The advisory members viewed the benefits of the Pfizer vaccine as greater than the potential risks, and except for one abstention, all voted in favor of recommending vaccination for children.
In a report released ahead of the meeting, FDA experts stated, "If the probability of severe side effects such as myocarditis in children aged 5 to 11 is similar to that in those aged 12 to 15, the preventive effect against severe COVID-19 infection will outweigh the risks caused by side effects."
Although the VRBPAC decision is strongly advisory, the FDA has typically accepted such external advisory committee decisions, increasing the likelihood of emergency use authorization for vaccination targeting children.
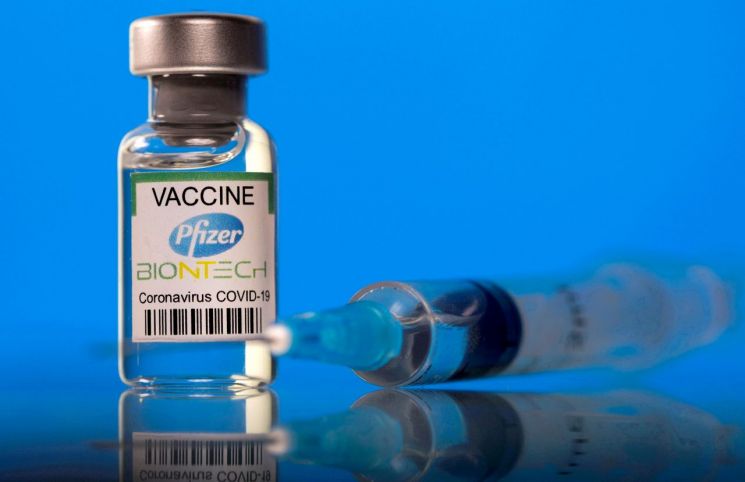
If the FDA grants emergency use authorization, actual vaccinations for these children are expected to begin early next month after additional approval procedures by the Centers for Disease Control and Prevention (CDC). The Pfizer vaccine dose for children aged 5 to 11 is reported to be 10μg, one-third of the adult dose (30μg).
Previously, Pfizer announced that its vaccine demonstrated 90.7% immune efficacy in children aged 5 to 11.
Children and adolescents have relatively lower rates of severe illness and fatality from COVID-19 infection compared to adults. However, recent concerns have arisen that unvaccinated children could die from complications due to COVID-19 infection influenced by the Delta variant.
Dr. Amanda Cohn, a member of the advisory panel participating in the vote and a vaccine expert at the CDC, emphasized, "We do not want to see children dying from COVID-19," adding, "They should not be admitted to intensive care units."
Currently, in the United States, Pfizer vaccine administration is available for those aged 12 and older. Moderna and Janssen vaccines are available for those aged 18 and older.
Moderna applied for emergency use authorization for ages 12 to 17 in June but has not yet been approved.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














