Overcoming Immune Checkpoint Signals to Expand Treatment Targets
Professor Kim Chanhyeok's Team at KAIST
[Asia Economy Reporter Kim Bong-su] The Korea Advanced Institute of Science and Technology (KAIST) announced on the 20th that Professor Kim Chan-hyuk’s research team from the Department of Biological Sciences has developed a next-generation chimeric antigen receptor T (CAR-T) cell therapy that overcomes immune checkpoint signals.
CAR-T cell therapy is a gene cell therapy that introduces the CAR gene into T cells, immune cells that play a crucial role in anti-cancer and antiviral functions in our body, enhancing their anti-cancer capabilities. It has shown a high therapeutic effect of over 80% in clinical trials targeting terminal leukemia patients who are refractory to all existing cancer treatments, earning it the nickname “miracle anticancer drug.”
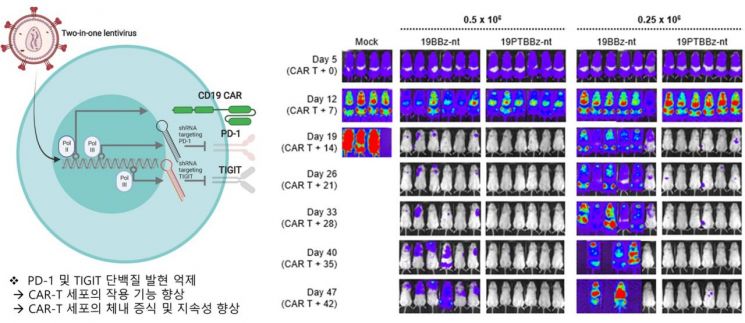
Professor Kim’s research team improved the lentiviral vector used to produce CAR-T cell therapy so that two types of short hairpin RNA (shRNA) are co-expressed with the CAR gene. By simultaneously suppressing the expression of two immune checkpoint receptors, PD-1 and TIGIT, which induce T cell dysfunction, they confirmed enhanced anti-cancer functions of CAR-T cells in leukemia and lymphoma models using mice.
The research results were licensed to Qurocell, a CAR-T cell therapy specialized venture co-founded by Professor Kim, and since March of this year, a phase 1b/2a clinical trial has been underway at Samsung Medical Center targeting patients with diffuse large B cell lymphoma (DLBCL) who relapse or are refractory after conventional cancer treatments. This is the first CAR-T clinical trial attempted with domestic technology in Korea.
Due to its high anticancer efficacy, two CAR-T therapies were first approved in the United States in 2017, and based on active research in academia and industry, a total of five CAR-T therapies have been approved to date. Recently, China has rapidly emerged as a new powerhouse in the CAR-T therapy field through large-scale investments and aggressive clinical research, with more than half of the approximately 500 ongoing CAR-T clinical trials worldwide currently being conducted in China. In contrast, only one clinical trial is currently underway in Korea.
Although CAR-T therapies have attracted much attention due to their high therapeutic effects, the dramatic clinical efficacy has so far been limited to hematologic cancers such as B cell acute leukemia and multiple myeloma. Among hematologic cancers, the therapeutic effect is relatively low in B cell chronic leukemia and lymphoma, and most importantly, there is still no CAR-T therapy showing high efficacy in solid tumors, which remains a critical challenge to be addressed.
The research team focused on immune checkpoint receptors that inhibit T cell activation, which are potential factors limiting CAR-T cell efficacy. Various immune checkpoint receptors expressed on T cells originally function to prevent side effects caused by continuous T cell activation, but it is well known that cancer cells exploit these to reduce T cell activity and evade immune system action.
Based on a platform that simultaneously expresses two types of shRNA, the team suppressed the expression of various combinations of immune checkpoint receptors and interestingly found that the combination of PD-1 and TIGIT uniquely enhanced CAR-T cell function. Subsequent transcriptome analysis and cell function tests revealed that while PD-1 suppression enhances the effector function of CAR-T cells, TIGIT suppression delays differentiation, thereby improving CAR-T cell proliferation and persistence in vivo.
Postdoctoral researcher Lee Young-ho said, “Blocking PD-1 and TIGIT signals is a novel technological strategy designed to help CAR-T cells overcome immune suppression, and it is expected to be an essential therapy for lymphoma patients who have difficulty benefiting from existing treatments.” He added, “The experience gained from developing CAR-T therapies will be a great asset for developing new therapies, including for solid tumors.”
This study was published in the October online edition of 'Molecular Therapy,' the official journal of the American Society of Gene & Cell Therapy.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














