[Asia Economy Reporter Kwon Jae-hee] The U.S. Food and Drug Administration (FDA) has begun reviewing the emergency use authorization for the oral COVID-19 treatment developed by Merck & Company (hereafter Merck). However, since the review will involve an external advisory committee, approval before December is expected to be difficult.
According to Bloomberg on the 15th (local time), the FDA decided to determine the emergency use authorization of Merck's oral COVID-19 treatment, Molnupiravir, through an external advisory committee review and plans to hold the advisory committee meeting on November 30.
The external advisory committee will evaluate the safety and efficacy of Molnupiravir at this meeting.
The FDA sometimes skips the external advisory committee review stage depending on the urgency of the situation.
However, due to some experts raising safety concerns regarding MSD's emergency use authorization application, the FDA decided to gather the advisory committee's opinions before making a final decision.
Opinions on the clinical data of Molnupiravir submitted by MSD will also be conveyed.
Considering that the first external advisory committee meeting is scheduled for November 30 and the FDA will review the committee's opinions before making a final decision, it seems unlikely that the first oral COVID-19 treatment will receive approval before December.
The FDA stated, "The external advisory committee review will help clearly understand the scientific data and information in deciding on the emergency use of Molnupiravir."
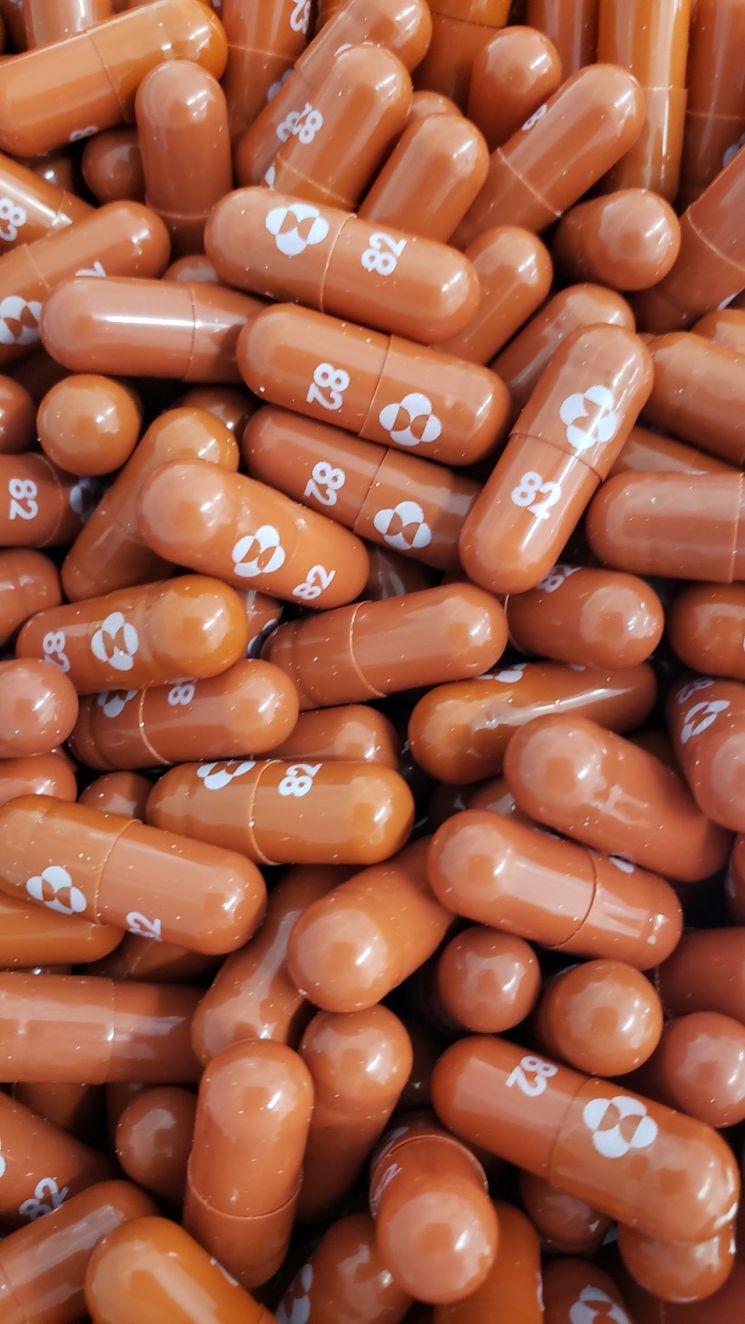
Molnupiravir is a treatment developed for adult COVID-19 patients who exhibit mild to moderate symptoms but are at high risk of progressing to severe illness.
MSD plans to produce 10 million doses by the end of this year. The U.S. federal government is reported to have signed a pre-purchase agreement for 1.7 million doses.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















