Russians "Oppose Vaccine Discrimination Attempts"
China's Sinopharm and Sinovac Vaccines Allowed
[Asia Economy Reporter Hyunwoo Lee] The U.S. health authorities are planning not to allow entry to those who have completed vaccination with Russia's COVID-19 vaccine, Sputnik V, sparking controversy. The U.S. health authorities plan to allow entry to those who have completed vaccination with vaccines that have received emergency approval from the U.S. Food and Drug Administration (FDA) and the World Health Organization (WHO), but Sputnik V has not yet received WHO approval and is expected to be excluded from the list of allowed entries. Russia has protested, demanding an end to vaccine discrimination.
According to the Washington Post (WP) on the 27th (local time), U.S. authorities plan not to allow entry to foreign nationals vaccinated with vaccines that have not received emergency use authorization from the FDA or WHO, including Russia's Sputnik V vaccine. As a result, all Russian visitors seeking to enter the U.S. are expected to be blocked immediately. According to the U.S. Travel Association (USTA), about 300,000 Russian visitors traveled to the U.S. in 2019.
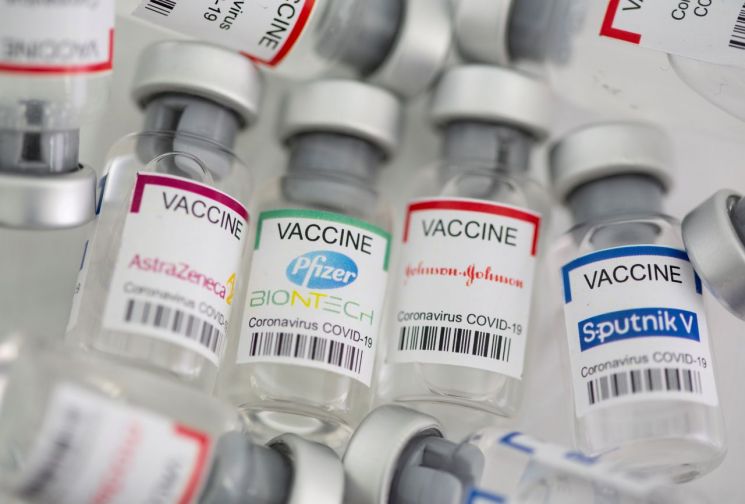
The vaccines currently accepted by U.S. authorities as entry criteria include Pfizer, Moderna, Janssen, AstraZeneca, as well as China's Sinopharm and Sinovac vaccines. These vaccines have received emergency use authorization from the FDA or WHO. However, Sputnik V has not yet received emergency approval from either organization. Additionally, WHO recently raised concerns about manufacturing practices at production facilities in Russia and announced the suspension of the emergency use approval review for Sputnik V.
Foreign nationals wishing to enter the U.S. are required to have completed vaccination with vaccines that have received emergency approval from the FDA or WHO, but Sputnik V, developed by the Gamaleya Institute under the Russian Ministry of Health, unlike Pfizer and Moderna developed by Western institutes or Sinopharm and Sinovac developed by Chinese companies, has not yet received WHO emergency approval.
Russian authorities have immediately protested. The Russian Direct Investment Fund (RDIF), responsible for the overseas production and supply of Sputnik V, stated in a press release, "Sputnik V has been approved in 70 countries where more than 4 billion people, over half of the world's population, reside, and its efficacy and safety have been confirmed through clinical trials and actual use in multiple countries." They emphasized, "We oppose the politicization of the global fight against COVID-19 and attempts to discriminate against effective vaccines for short-term political and economic gains."
Sputnik V was the world's first COVID-19 vaccine developed and approved by Russia in August last year, bypassing the usual vaccine development procedures. It was approved based on clinical trials involving only 28 subjects by combining phase 1 and 2 results before phase 3 clinical trials, leading to ongoing controversy over the vaccine's efficacy and safety.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














