Production Technology Research Institute Develops Surface Stabilization Technology for Aqueous Zinc Secondary Battery Anodes
Maintains 93% Capacity After 3000 Charges, Successfully Achieves Large Capacity
Next-Generation Secondary Battery to Replace Fire-Prone Lithium-Ion Batteries
[Asia Economy Reporter Kim Bong-su] On the 15th, the Korea Institute of Industrial Technology announced that it has developed an anode surface stabilization technology for aqueous zinc secondary batteries, emerging as the next-generation secondary battery, successfully solving the biggest obstacle to commercialization?the battery lifespan degradation issue.
Recently, with the government's Green New Deal policy promotion, renewable energy power generation facilities such as solar and wind power have been increasing by more than 10% annually. However, due to output instability caused by weather conditions, an Energy Storage System (ESS) is essential for stable power supply. Currently, most ESS installations in Korea use lithium-ion batteries. The problem is that dozens of ESS fire accidents have continuously occurred over the past four years, raising safety concerns about secondary batteries.
Aqueous zinc secondary batteries are attracting attention as the next-generation secondary battery that can succeed lithium-ion batteries, which have frequent fire accidents. Since they use water-based electrolytes, there is no risk of ignition, and they offer high stability. Cathode materials can be synthesized without high-temperature heat treatment, and batteries can be assembled in normal atmospheric conditions rather than in a dry room.
However, because zinc metal is used as the anode, corrosion occurs in the water-based electrolyte, and especially zinc ions tend to accumulate sharply on the anode surface in the form of branch-like crystals. These crystals, called “dendrites,” hardly participate in charge-discharge reactions but continue to grow. If they penetrate the separator and contact the cathode, they eventually cause short circuits, drastically reducing battery life and triggering fires.
The research team was the first in the world to observe through electron microscopy that dendrite formation is suppressed and its morphology changes depending on the chemical properties of the zinc anode surface. Specifically, they found that the more hydrophilic the zinc anode surface is?meaning it easily bonds with water molecules?the more uniformly zinc ions adsorb onto the anode surface during battery charging, suppressing dendrite formation. Conversely, on hydrophobic anode surfaces, zinc ion distribution concentrates in relatively less hydrophilic areas, resulting in dendrites shaped like spheres tens of micrometers (㎛) in size forming sporadically.
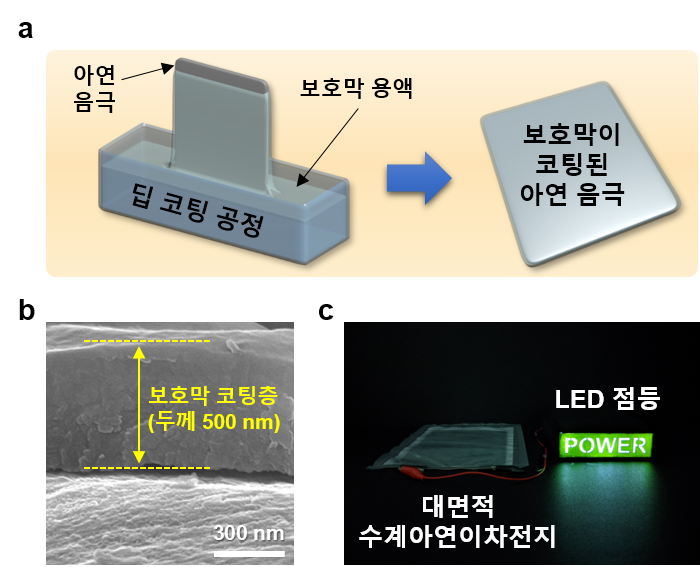
Furthermore, the team succeeded in effectively suppressing dendrite formation and corrosion reactions by uniformly forming a thin 500-nanometer (㎚) thick hydrophilic protective film on the anode surface through a simple dip-coating process. The dip-coating process involves immersing the anode material in a coating solution to form a layer, then heating it to create a protective film, which is advantageous for continuous production processes.
As a result of forming the anode protective film using this method, the battery demonstrated stable lifespan characteristics with a capacity retention rate of 93% even after about 3,000 harsh charge-discharge cycles. Additionally, the self-discharge rate, which is the rate at which charging power naturally dissipates, was suppressed more than twice compared to uncoated anodes. While previous studies on zinc anode protection were limited to laboratory-level experiments on coin cells the size of a fingernail, this study differentiated itself by demonstrating the formation of a protective film on a large-area (176㎠) zinc anode more than 150 times larger using a simple process, proving its mass production potential.
Dr. Kim stated, “This research has solved the technical challenges of aqueous zinc batteries and increased their commercialization potential.” The research results were published online on the 10th in the international energy journal ‘ACS Energy Letters (IF=23.101)’.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














