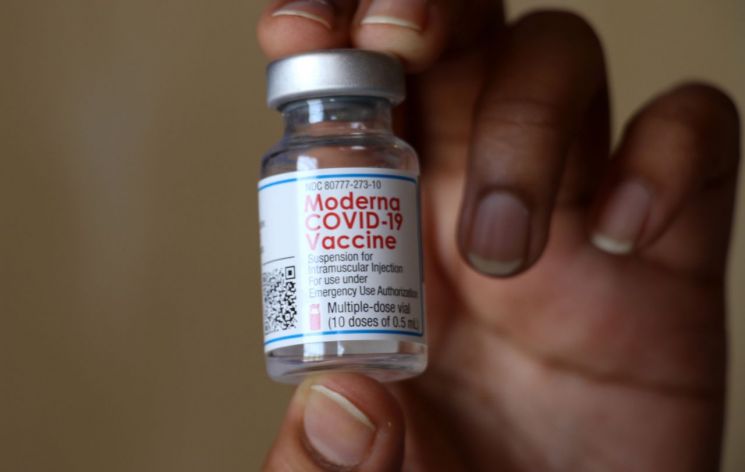
[Asia Economy Reporter Lee Chun-hee] Regulatory authorities have begun the review process to lower the eligible age for Moderna's COVID-19 vaccine to 12 years old.
On the 27th, the Ministry of Food and Drug Safety announced that GC Green Cross had applied for a license amendment to expand the eligible age for the "Moderna COVID-19 vaccine" from the existing 18 years and older to 12 years and older.
Green Cross submitted clinical trial results conducted in the United States on 3,732 adolescents aged 12 to 17 as the basis for this license amendment application. The clinical trial involved comparing immunogenicity and confirming the effectiveness in preventing COVID-19 infection and safety. At that time, Moderna announced that no COVID-19 infections occurred in the vaccinated group, while four infections occurred in the placebo group. Earlier, the European Medicines Agency (EMA) approved the vaccination of adolescents aged 12 to 17 with the Moderna vaccine on the 23rd.
If this license amendment is approved, the Moderna vaccine will become the second vaccine available for minors in Korea after Pfizer among the vaccines currently introduced domestically. On the 16th, the Ministry of Food and Drug Safety lowered the eligible age for Pfizer's COVID-19 vaccine "Comirnaty" from 16 to 12 years and approved the amendment. Currently, both Pfizer and Moderna are conducting trials in the United States to determine whether COVID-19 vaccination is possible for children under 11 years old.
A Ministry of Food and Drug Safety official stated, "We plan to review this amendment application quickly and thoroughly."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














