Korea Institute of Oriental Medicine Plans to Confirm Treatment Effectiveness for Colorectal Cancer Patients with Standard Therapy Failure
[Asia Economy Reporter Kim Bong-su] An anticancer drug made from herbal medicine ingredients has attracted attention as it has entered phase 2 clinical trials for the first time ever.
The Korea Institute of Oriental Medicine announced on the 23rd that it received approval from the Ministry of Food and Drug Safety on the 3rd of this month for the phase 2 clinical trial plan of ‘KIOM-ICI-1,’ an immune checkpoint inhibitor candidate based on Korean medicine. Immune anticancer drugs are treatments that either prevent cancer cells from evading the immune system or enhance immune cells’ ability to attack cancer cells, including immune checkpoint inhibitors, cancer vaccines, and immune cell therapies.
This approval for the phase 2 clinical trial plan is the first case where immune anticancer treatment efficacy blocking immune checkpoints, rather than antibody therapy, was discovered in a material based on herbal medicine, leading to clinical trial approval. Unlike conventional treatments that attack cancer cells directly, immune anticancer therapies improve immune function to treat cancer and are gaining attention as next-generation anticancer treatments. Among them, immune checkpoint inhibitors are the most widely used immune anticancer drugs, with seven products currently approved; however, due to low response rates (2 out of 10 patients) and side effects such as immune hypersensitivity reactions, there is a need to discover new materials to complement these issues.
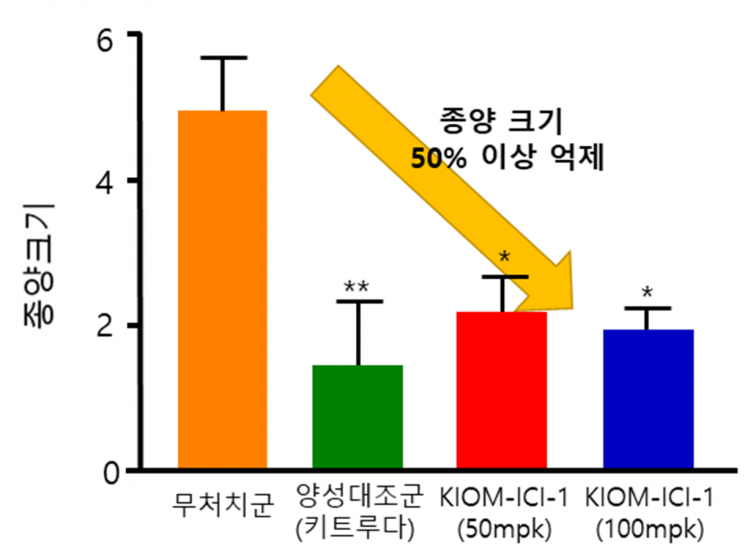
The research team plans to conduct a randomized clinical trial targeting colorectal cancer patients who have failed standard anticancer treatments such as surgery, chemotherapy, and radiation therapy, to verify the safety and efficacy of the immune checkpoint inhibitor ‘KIOM-ICI-1.’ Previously, the team confirmed that ‘KIOM-ICI-1’ blocks the immune checkpoint, a mechanism by which cancer cells evade the immune system, and enhances the activity of immune cells (T-cells), suppressing tumor size by more than 50%. They also found that the therapeutic effect of ‘KIOM-ICI-1’ increases when administered in combination with the existing cytotoxic anticancer drug oxaliplatin, and plan to conduct clinical trials on combination therapy with existing anticancer drugs in the future.
Jung Hwan-seok, principal researcher at the Korea Institute of Oriental Medicine, said, “This anticancer immune candidate material is significant as it was developed based on herbal medicine with proven safety,” adding, “We will prepare follow-up research to develop KIOM-ICI-1 as the first Korean medicine-based immune anticancer drug.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















