[Asia Economy Reporter Kwon Seoyoung] In Buan-gun, Jeonbuk, an incident occurred where an excessive dose of the Janssen vaccine was administered, drawing attention to the background of the situation.
On the 12th, Jeonbuk health authorities announced that the Buan-gun public health center received a report of a man in his 30s who had received the Janssen vaccine experiencing a high fever of around 40 degrees Celsius. Upon investigation, the health authorities confirmed that a private commissioned medical institution had administered more than five times the recommended dose of the Janssen vaccine to five men in their 30s. These individuals were subsequently transferred to a hospital, and except for one who showed high fever symptoms, no other adverse reactions were observed among the others.
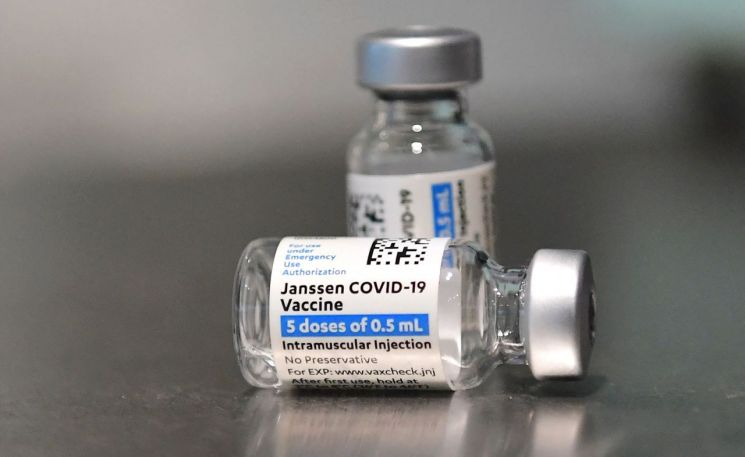
The recommended dose of the Janssen vaccine is 0.5ml, and one vial (3ml) should be divided and administered to 5 to 6 people. However, it was found that the medical staff at the hospital where the incident occurred administered an entire vial of the Janssen vaccine to a single person. The cause of the incident is currently presumed to be due to negligence on the part of the medical staff.
AstraZeneca (AZ) and Pfizer vaccines use special syringes during administration. These syringes have a smaller capacity than regular syringes, making overdosing incidents virtually impossible. However, unlike the AZ or Pfizer vaccines, the Janssen vaccine has not been supplied with dedicated syringes. This means that medical staff must use regular syringes with a maximum capacity of 3 to 5ml when administering the Janssen vaccine. Consequently, there are criticisms that the hospital may not have paid sufficient attention to the syringe capacity.
Jeonbuk health authorities have determined that it is impossible for the medical institution in question to continue vaccination and will proceed with canceling its private commissioned medical institution status. Additionally, an official stated, "No such incidents have occurred at other medical institutions in similar situations," and "We are investigating the exact circumstances with the clinic involved, as this was an accident that should never have happened," adding, "If any illegalities are found, appropriate measures will be taken."
Meanwhile, earlier in Incheon, controversy arose when it was revealed that a hospital administered about half the recommended dose of the AZ vaccine to around 40 recipients. The U.S. Centers for Disease Control and Prevention (CDC) stipulates that if less than the recommended dose is administered, the decision to revaccinate depends on the amount given, but if more than half the dose is administered, revaccination is generally not required.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















