<명령>Mandatory Disclosure of Product Information on Rare Thrombotic Side Effects
Occurs Within 3 Weeks Post-Vaccination in Under 60s... Mostly Women
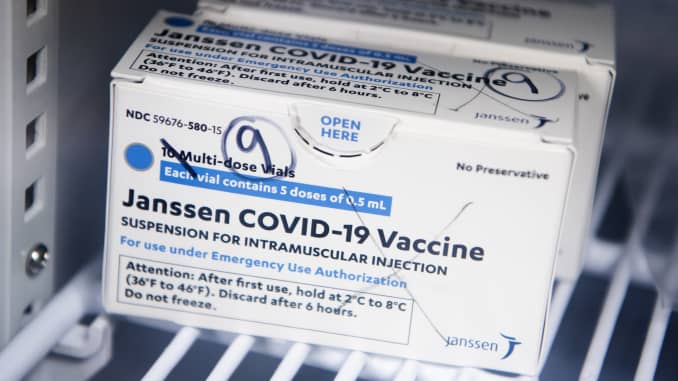
[Asia Economy Reporter Yujin Cho] On the 20th (local time), the European Medicines Agency (EMA) announced that the Janssen vaccine by the American company Johnson & Johnson (J&J) may be linked to symptoms of blood clot side effects.
The EMA's Safety Committee stated in a press release that day, "We have found a possible link between the Janssen vaccine and very rare cases of unusual blood clots accompanied by low platelet counts."
However, the EMA said, "The benefits of receiving the Janssen vaccine outweigh the risks of side effects," and decided to maintain the authorization of the Janssen vaccine while ordering that the very rare side effect of unusual blood clots accompanied by low platelet counts be included in the product information of the Janssen vaccine.
The EMA previously took a similar stance in its evaluation of the AstraZeneca (AZ) vaccine by the multinational pharmaceutical company.
EMA Executive Director Emer Cook said at a press conference that day, "I want to emphasize once again that these cases are very rare, and in most cases, these vaccines will prevent death and hospitalization due to COVID-19."
The EMA explained that in reaching this conclusion, it reviewed all available evidence, including reports of eight serious cases of unusual blood clots associated with low platelet counts reported in the United States. As of the 13th of this month, more than 7 million people in the U.S. have received the Janssen vaccine.
All cases occurred within three weeks after vaccination in individuals under 60 years old, with the majority being women.
The EMA added that the reviewed cases were very similar to those that occurred in relation to the AstraZeneca (AZ) vaccine by the multinational pharmaceutical company.
However, Sabine Strauss, Chair of the EMA Safety Committee, said at the press conference that it is still too early to draw conclusions about the similarities between the two vaccines.
The EMA emphasized that healthcare workers and those scheduled to receive the vaccine should be aware of these very rare cases and that immediate treatment is crucial.
Cook stated, as reported by dpa news agency, that it is important for doctors and patients to recognize the signs related to this issue so that expert help can be sought as soon as possible, and that early intervention by experts can change outcomes.
The EMA said it will continue to analyze new data and evidence and provide updated guidelines if necessary.
Earlier, the U.S. Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC) recommended halting the use of the Janssen vaccine on the 13th, based on six cases of "rare but serious" blood clots among Janssen vaccine recipients.
Since then, many countries around the world, including the U.S., have suspended or postponed the introduction of this vaccine.
The first shipments of the Janssen vaccine began arriving in EU member states on the 12th, but some member states have temporarily paused vaccination with this vaccine while awaiting the EMA's evaluation results.
The EMA stated that each member state will design and implement its vaccination plans based on EMA recommendations, which may vary depending on each country's infection rates, available vaccines, and other circumstances.
Previously, on the 7th, the EMA Safety Committee concluded that unusual blood clots accompanied by low platelet counts should be included as a very rare side effect of the AZ COVID-19 vaccine.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














