A total of 79 million vaccine doses planned for introduction
High distrust in AstraZeneca due to 'blood clot controversy'
Janssen also faces blood clot issues
Novavax and Moderna face supply instability
 On the morning of the 1st, a citizen is receiving the Pfizer vaccine at the COVID-19 local vaccination center set up at Yeomju Gymnasium in Seo-gu, Gwangju. [Image source=Yonhap News]
On the morning of the 1st, a citizen is receiving the Pfizer vaccine at the COVID-19 local vaccination center set up at Yeomju Gymnasium in Seo-gu, Gwangju. [Image source=Yonhap News]
[Asia Economy Reporter Lee Chun-hee] As a series of setbacks have emerged regarding vaccines scheduled for domestic introduction, doubts are growing about the government's plan to achieve 'herd immunity by November.' Following the AstraZeneca vaccine, the main vaccine to be introduced domestically, concerns about blood clots have also arisen with the Janssen vaccine, which is scheduled for 6 million doses, and all other vaccines are suffering from supply instability.
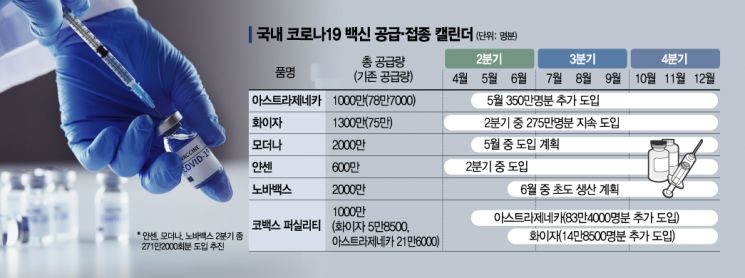
According to health authorities on the 14th, the total amount of vaccines scheduled for domestic introduction is 79 million doses. By product, the plan is to introduce △AstraZeneca 10 million doses (20 million shots) △Pfizer 13 million doses (26 million shots) △Moderna 20 million doses (40 million shots) △Novavax 20 million doses (40 million shots) △Janssen (a subsidiary of Johnson & Johnson) 6 million doses, plus 10 million doses from the global vaccine joint purchase project 'COVAX Facility.'
Vaccines Confirmed for Introduction in the First Half: Only AstraZeneca and Pfizer
Looking at the period, the amount with a confirmed specific introduction schedule in the first half is 9.044 million doses (18.088 million shots). AstraZeneca accounts for 5.337 million doses (59%), and Pfizer 3.707 million doses (41%). Excluding the doses already introduced, plans are in place to introduce 4.334 million doses of AstraZeneca and 2.8985 million doses of Pfizer. Regarding this, Kwon Deok-cheol, Minister of Health and Welfare, stated yesterday that "20.8 million shots of vaccines will be introduced in the first half." An additional 2.712 million shots are reportedly under further discussion. The task force is negotiating the introduction of Janssen, Moderna, and Novavax doses.
In fact, until the first half, vaccinations for the scheduled 12 million people with AstraZeneca and Pfizer vaccines must be carried out, but the vaccination schedule is facing more setbacks as red flags continue to be raised about the safety of AstraZeneca, which is the most critical. Initially, safety concerns for the elderly led to excluding those aged 65 and over from the vaccination target, then they were included again, and now, with the confirmed possibility of thrombosis, 640,000 people under 30 have been excluded from AstraZeneca vaccination targets.
The government plans to maintain the overall vaccination level without disruption, but the problem is public distrust. The elderly have significant distrust of AstraZeneca due to earlier safety concerns, and distrust is also growing among people in their 30s, who narrowly remained eligible for AstraZeneca vaccination. Office worker Kim Sang-jin (32) said, "What's the difference between people in their 20s and early 30s?" and added, "If I become a target for AstraZeneca vaccination, I will postpone it."
Blood Clot Side Effect Controversy Arises with Janssen Following AstraZeneca
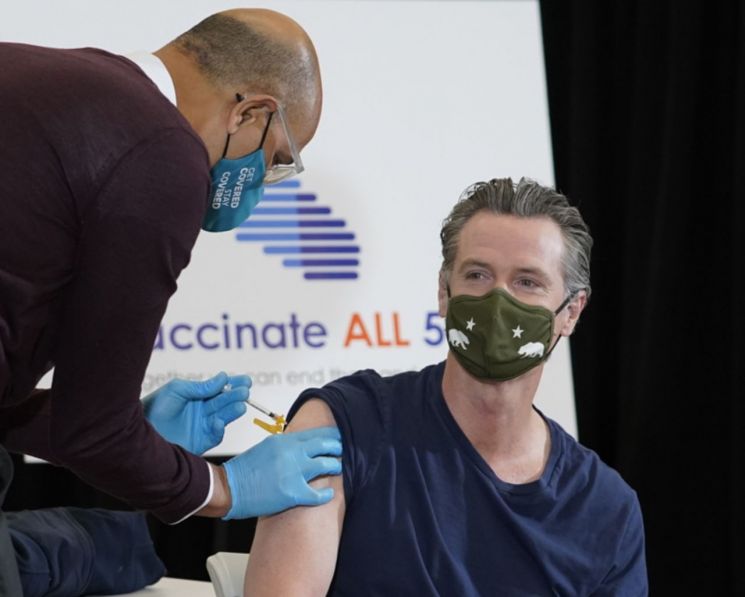 On the 1st (local time), Gavin Newsom, Governor of California, USA, received the Janssen (a subsidiary of Johnson & Johnson) COVID-19 vaccine.
On the 1st (local time), Gavin Newsom, Governor of California, USA, received the Janssen (a subsidiary of Johnson & Johnson) COVID-19 vaccine. [Image source=AP Yonhap News]
The problem is that Janssen, Novavax, and Moderna vaccines, excluding Pfizer, are also facing continuous obstacles in safety and supply. On the 13th (local time), the U.S. Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC) recommended halting the use of the Janssen vaccine after blood clots were found in six vaccinated individuals. Vaccinations with Janssen have been suspended not only in the U.S. but also in South Africa, and Johnson & Johnson announced a delay in vaccine launch in Europe.
The Novavax vaccine, which the government announced would be supplied "as early as May," is delayed due to improper supply of raw and subsidiary materials. Production is expected to start as early as June, and all 10 million doses (20 million shots) planned to be produced by SK Bioscience in the third quarter will be introduced domestically. However, the fact that there are no global cases of use yet is considered a risk factor.
The Moderna vaccine was announced by the company at the end of last year to be planned for supply in Korea starting in May, but no further plans have been announced since then.
The government has not yet disclosed specific plans for vaccine introduction volumes by pharmaceutical companies in the second quarter. Baek Young-ha, head of the government-wide vaccine introduction TF vaccine introduction general team, said at a briefing this morning, "Since it is still at a stage where nothing is specifically confirmed, we cannot provide details."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














