[Asia Economy Reporter Lee Chun-hee] Pfizer's COVID-19 vaccine has smoothly passed the first hurdle for official domestic product approval. It showed about 95% preventive efficacy, and opinions were given that administration to adolescents aged 16-17 is also appropriate.
The Ministry of Food and Drug Safety (MFDS) held a meeting of the "COVID-19 Vaccine Safety and Effectiveness Verification Advisory Committee" on the 22nd regarding the clinical trial results of Korea Pfizer's COVID-19 vaccine "Comirnaty" and announced these results on the 23rd. Before approving COVID-19 vaccines and treatments, the MFDS conducts a three-layered advisory review process involving the verification advisory committee, the Central Pharmaceutical Affairs Deliberation Committee, and the final inspection committee. Seven experts, including infectious disease specialists, vaccine experts, and clinical statistics experts, attended this verification advisory committee meeting.
This verification advisory was based on clinical trial results conducted in six countries, including Germany and the United States. A total of 43,448 people received at least one dose of the vaccine or control drug, with 41.1% (17,846 people) aged 55 or older included.
According to the verification advisory committee's analysis, Pfizer showed over 94% effectiveness in preventing COVID-19. Among 36,523 people who had not been confirmed with COVID-19 infection within 7 days after two doses of vaccine or control drug, 8 out of 18,198 in the vaccine group and 162 out of 18,325 in the control group were confirmed COVID-19 cases. The resulting preventive efficacy was about 95%, showing consistent preventive effects of over 94% regardless of age or underlying health conditions.
However, regarding prevention of severe COVID-19, the verification advisory committee analyzed that although there was a trend of prevention with one severe case in the vaccine group and three in the control group, the number of cases was too small to have statistical significance.
Additionally, Pfizer's vaccine received high evaluations in immunogenicity assessments, which evaluate immune responses such as the types and amounts of antibodies produced in the body after vaccination, serving as indirect indicators to predict vaccine effectiveness. For "binding antibodies" that bind to the COVID-19 virus antigen, antibody titers against the antigen increased more than fourfold in all subjects compared to before vaccination. Accordingly, the "seroconversion rate," indicating the proportion of subjects whose antibody titers increased fourfold or more, was 100%.
Furthermore, "neutralizing antibodies," which bind to the virus particle surface and neutralize the virus's infectivity to induce preventive effects, also showed antibody titers increased more than fourfold in all subjects after two vaccine doses, resulting in a seroconversion rate of 100%.
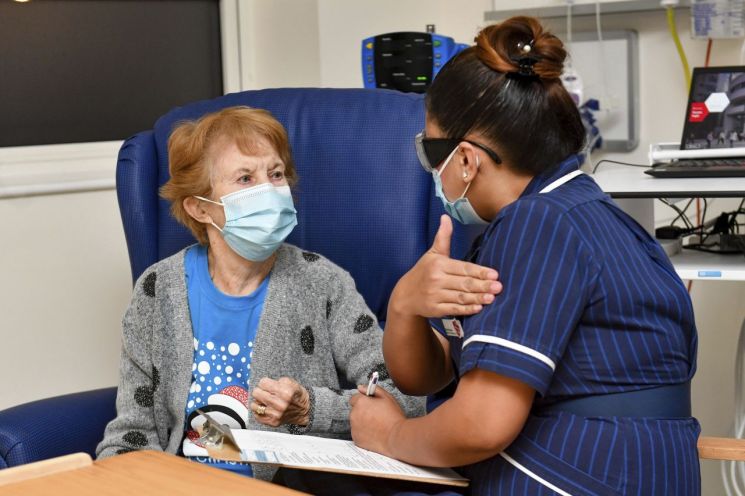 In December last year, Margaret Keenan (90), the world's first general recipient of the COVID-19 vaccine, is listening to a nurse's explanation before receiving the Pfizer vaccine. [Image source=AP Yonhap News]
In December last year, Margaret Keenan (90), the world's first general recipient of the COVID-19 vaccine, is listening to a nurse's explanation before receiving the Pfizer vaccine. [Image source=AP Yonhap News]
Regarding safety, among 37,586 people monitored for 4 weeks after vaccination, unexpected adverse events related to vaccination occurred in 20.8% of the vaccine group. These included ▲injection site pain 11.2%, ▲fever 6%, and ▲fatigue 5.3%. One case of drug-related hypersensitivity (urticaria) was reported, and no anaphylaxis was reported among clinical trial institutions.
The verification advisory committee judged the safety profile (trend) to be at an acceptable level but recommended careful monitoring for individuals with a history of anaphylaxis.
The verification advisory committee also evaluated that Pfizer's COVID-19 vaccine is suitable for administration to adolescents aged 16-17. Previous clinical results assessing preventive efficacy (107 subjects) and safety (283 subjects) showed no COVID-19 infection cases in either the vaccine or control groups, and adverse events were similar to those in other age groups.
Accordingly, the verification advisory committee concluded that it is appropriate to approve vaccination for adolescents aged 16-17, considering ▲clinical trials designed to confirm safety and efficacy in subjects aged 16 and older with confirmed preventive efficacy, ▲the immune response of 16-17-year-old adolescents is expected to be similar to adults, ▲availability of adult clinical trial data, and ▲approval for ages 16 and older in multiple countries including the United States, Europe (EU), the United Kingdom, and Japan.
 On the 22nd, medical staff are moving at the COVID-19 Central Vaccination Center set up at the National Medical Center in Jung-gu, Seoul. COVID-19 vaccinations are scheduled to begin on the 26th. Photo by Mun Ho-nam munonam@
On the 22nd, medical staff are moving at the COVID-19 Central Vaccination Center set up at the National Medical Center in Jung-gu, Seoul. COVID-19 vaccinations are scheduled to begin on the 26th. Photo by Mun Ho-nam munonam@
Finally, the verification advisory committee judged that the preventive efficacy of Pfizer's COVID-19 vaccine is sufficient, as effectiveness was confirmed in the submitted data and immune responses were maintained at levels higher than antibodies in convalescent plasma from natural infection. They also expressed the opinion that it is appropriate to continuously monitor safety, including anaphylaxis, through a post-approval 'Risk Management Plan' and to continuously collect and evaluate information on adverse events observed during clinical trials.
The MFDS is conducting a review of quality data related to Comirnaty and plans to receive advisory opinions from the Central Pharmaceutical Affairs Deliberation Committee on the 25th and disclose the results on the 26th, synthesizing expert opinions, recommendations from this verification advisory committee, and review results.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














