Government "Introduction Before Lunar New Year"
AstraZeneca Proceeding with Product Approval Process
Pfizer, Special Import for 'COVAX' Introduction
 On the 21st, market merchants are undergoing COVID-19 diagnostic tests at a temporary walk-in screening clinic set up in Namdaemun Market, Jung-gu, Seoul. Jung-gu explained, "Since it is difficult for merchants to leave their shops during business hours due to sales and other operations, we collaborated with Seoul City to establish a testing site inside the market for merchants who cannot visit screening clinics." Photo by Hyunmin Kim kimhyun81@
On the 21st, market merchants are undergoing COVID-19 diagnostic tests at a temporary walk-in screening clinic set up in Namdaemun Market, Jung-gu, Seoul. Jung-gu explained, "Since it is difficult for merchants to leave their shops during business hours due to sales and other operations, we collaborated with Seoul City to establish a testing site inside the market for merchants who cannot visit screening clinics." Photo by Hyunmin Kim kimhyun81@
[Asia Economy Reporter Lee Chun-hee] As the government has suggested the possibility of introducing a COVID-19 vaccine before the Lunar New Year, attention is focusing on which vaccine will be the first to be administered domestically.
According to the medical community and quarantine authorities on the 22nd, South Korea currently has secured a total of 56 million doses of vaccines, including AstraZeneca (10 million doses), Janssen (6 million doses), Pfizer (10 million doses), and Moderna (20 million doses). Negotiations are also underway to secure an additional 20 million doses from the U.S. company Novavax.
Among these, the vaccine initially expected to be introduced the earliest was AstraZeneca’s vaccine, as it is currently the only vaccine undergoing the government’s product approval process.
The Ministry of Food and Drug Safety (MFDS) plans to hold a verification advisory committee meeting on the AstraZeneca vaccine on the 31st. Before approving COVID-19 vaccines and treatments, the MFDS conducts a three-tier advisory review process including ▲verification advisory committee ▲central pharmaceutical review committee ▲final inspection committee. The approval decision is expected to be made within next month.
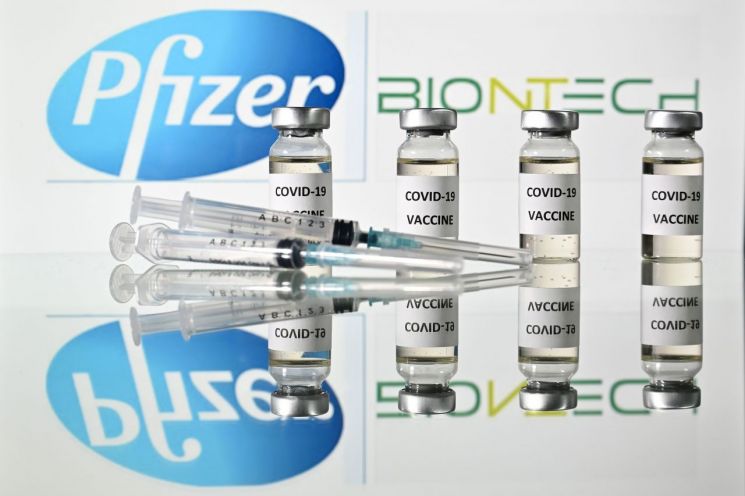
However, recently the possibility of Pfizer becoming the first vaccine in South Korea is gradually increasing. Although initially scheduled for introduction in the third quarter, the vaccine to be supplied next month through the international vaccine procurement and distribution project ‘COVAX Facility’ is narrowing down to Pfizer’s vaccine.
Prime Minister Chung Sye-kyun’s visit on the 20th to a production site of ultra-low temperature freezers essential for the ultra-cold 'cold chain' distribution below minus 70 degrees Celsius supports this outlook. Pfizer’s vaccine must be distributed through the cold chain to maintain its efficacy. Although this imposes a heavy burden during distribution, its preventive effect is known to be 95%, higher than AstraZeneca’s 70%.
However, even if Pfizer’s vaccine is introduced immediately, it is uncertain whether the government can meet its targeted vaccination schedule. Unlike AstraZeneca’s vaccine, Pfizer’s vaccine is still in the preliminary review stage before the product approval examination. Korea Pfizer stated, "We plan to apply for product approval from the MFDS as soon as possible."
Accordingly, the industry expects that a ‘special import’ procedure may be applied to Pfizer’s vaccine to meet the vaccination schedule. This system allows the import of medicines not yet approved domestically in response to crisis situations. If related ministries such as the Korea Disease Control and Prevention Agency request the MFDS and special import is approved, importation is possible without product approval. This was also applied when Gilead Sciences’ remdesivir, a COVID-19 treatment, was introduced in June last year.
At that time, the government explained, "We considered that remdesivir was allowed for use in the COVID-19 pandemic situation in the U.S., Japan, and the U.K." Currently, Pfizer’s vaccine is being administered in the U.K., U.S., Canada, Saudi Arabia, Israel, and other countries.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














