[Asia Economy Reporter Cho Hyun-ui] Chong Kun Dang announced on the 17th that it has signed an export contract for the biosimilar anemia treatment drug 'Nesvel' with Lotus, which is responsible for the Asia region of the multinational pharmaceutical company Alvogen.
Under this contract, Chong Kun Dang will supply finished Nesvel products to Lotus and receive an upfront payment and milestone payments (stage-based royalties). Lotus will conduct clinical trials and regulatory approval procedures for Nesvel in three Southeast Asian countries: Taiwan, Vietnam, and Thailand, and will have exclusive sales rights in these regions. The specific contract amount will not be disclosed according to the agreement between the two companies.
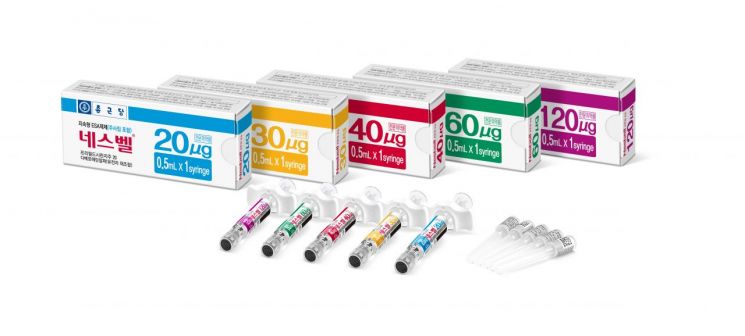
Nesvel is a biosimilar of 'Nesp,' an anemia treatment drug whose main ingredient is darbepoetin alfa, a long-acting erythropoiesis-stimulating agent. It is mainly used to treat anemia in patients with chronic renal failure. Using genetic engineering technology, it significantly reduces the frequency of drug administration, improving patient convenience as a second-generation long-acting product, and was launched last year in Korea and Japan.
A Chong Kun Dang official said, "With this contract, Nesvel has entered Southeast Asia following Japan," adding, "We plan to target the Nesp market worth 2.7 trillion KRW in Europe, the United States, and other regions in the future."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














