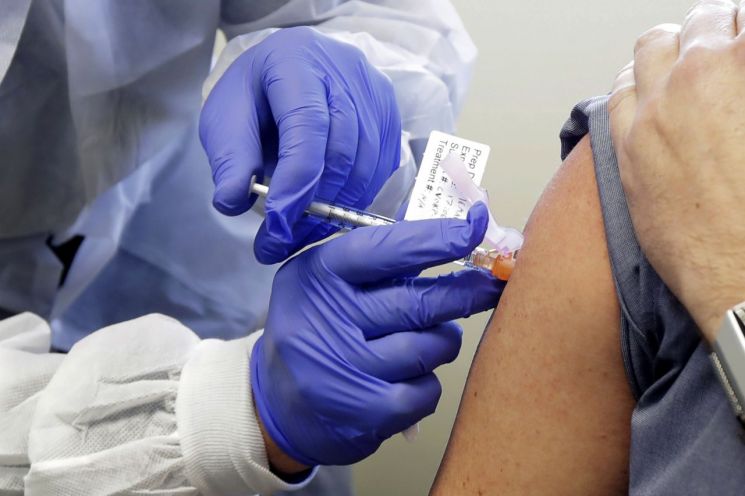
[Asia Economy Reporter Hyunwoo Lee] American pharmaceutical company Johnson & Johnson announced that it will begin Phase 3 clinical trials of its novel coronavirus disease (COVID-19) vaccine with 60,000 participants starting in September. The clinical trial involving 60,000 participants is the largest COVID-19 vaccine clinical trial to date. Johnson & Johnson expects that if this Phase 3 trial is successful, vaccine production could begin as early as the end of the year, allowing for emergency vaccine deployment from early next year.
According to foreign media such as CNBC, on the 20th (local time), Johnson & Johnson stated that it will start Phase 3 clinical trials of the COVID-19 vaccine from September, to be conducted at up to 180 sites worldwide, including the United States, with a maximum of 60,000 participants. Depending on the experimental group, participants will receive either the vaccine or a placebo, and the vaccine’s efficacy and safety will be verified through comparative analysis. Jake Sargent, a spokesperson for Johnson & Johnson, said, "Our Phase 3 trial will be conducted in areas with high infection rates."
Johnson & Johnson’s Phase 3 trial is expected to be the largest among COVID-19 vaccine clinical trials. Last month, U.S. biotech company Moderna and major pharmaceutical company Pfizer each began Phase 3 trials with 30,000 participants, which is about half the size of Johnson & Johnson’s trial. Earlier, British company AstraZeneca, which also announced the start of its Phase 3 trial, said it would conduct trials with about 47,000 participants worldwide, including 30,000 in the United States.
Johnson & Johnson stated that if the Phase 3 trial is successful, vaccine production will begin as early as the end of the year, and emergency vaccine deployment could be possible by early next year. They expect to increase production afterward, aiming to produce approximately 600 million to 900 million doses by April next year. Earlier, the U.S. Department of Health and Human Services announced on the 5th that it had signed a contract with Johnson & Johnson to purchase 100 million doses of the vaccine for about 1 billion dollars. The U.S. government also plans to have an option to order an additional 200 million doses in the future.
It is expected that from as early as the end of this year to early next year, pharmaceutical companies will competitively begin vaccine production. According to the World Health Organization (WHO), more than 160 vaccine development projects are underway worldwide, with at least 30 of them having entered clinical trial stages.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














