US Pharmaceutical Company Initiates Phase 3 Trial of Antiviral Drug
[Asia Economy Reporters Choi Daeyeol, Kim Heungsun] Domestic patients are expected to be included in clinical trials for the development of a treatment for the novel coronavirus infection (COVID-19). Currently, no COVID-19 treatment has been released anywhere in the world, but the pharmaceutical company developing one has decided to conduct clinical trials in Asian countries including South Korea. Research is also underway domestically to find drugs to apply for treatment, focusing on existing medications.
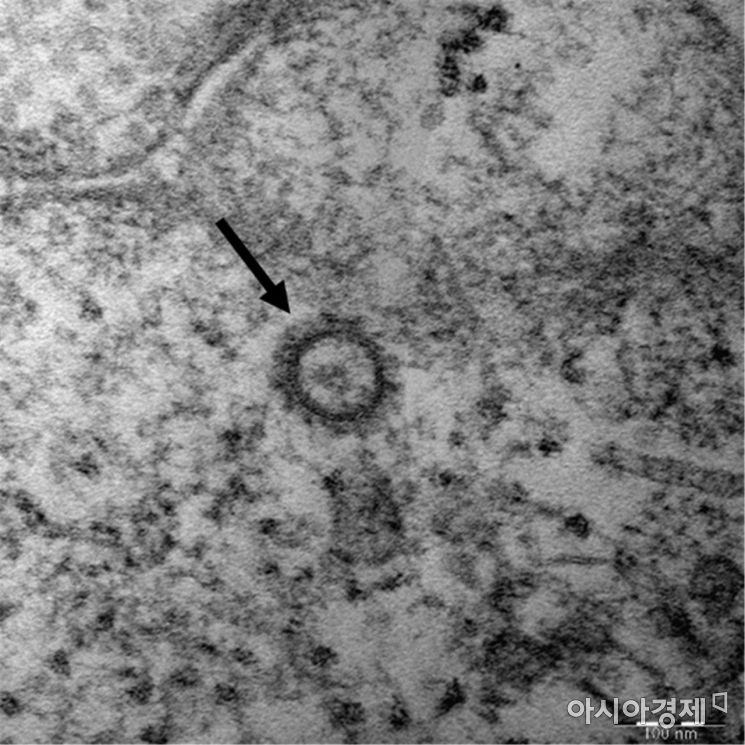
According to industry sources on the 28th, US pharmaceutical company Gilead Sciences has decided to initiate two Phase 3 clinical trials to study the efficacy and safety of 'Remdesivir' in confirmed COVID-19 patients. Remdesivir is an antiviral drug originally developed with the Ebola virus in mind, and this is the first Phase 3 clinical trial conducted on actual patients. Previously, the US Food and Drug Administration (FDA) granted expedited approval for the clinical trial plan, and the company has applied for clinical trial approval from regulatory authorities in countries including South Korea, China, and Japan.
Currently, HIV treatments such as Kaletra are being used on a limited basis for COVID-19 patients. It is difficult to draw clear conclusions about whether existing antiviral drugs are suitable for treating COVID-19, and diverse opinions remain within the medical community. Remdesivir is known to have shown efficacy, such as symptom improvement within a short period, based on its use in one patient in the US. The company is preparing a clinical trial involving 1,000 patients.
In South Korea, research led by the Korea Pasteur Institute is already underway to find drugs that can be applied to COVID-19 treatment among those that have been commercialized or whose safety has been verified. Research that discovers new drug effects from existing drugs is called 'drug repositioning.' According to related industries, developing a drug from candidate material and verifying its efficacy and safety takes over 10 years and astronomical costs, but drug repositioning can skip preclinical and clinical phases, thereby reducing development time and costs.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














